DMH1 in Guided Differentiation: Imaging and Tracking of hiPSC-Derived Intestinal Organoids
ABSTRACT
Using our reporter system to screen for hIO differentiation factors, we identified DMH1 as an efficient substitute for Noggin. Transplanted hIOs under the kidney capsule were tracked with fluorescence imaging (FLI) and confirmed histologically. Human intestinal organoids (hIOs) derived from human pluripotent stem cells (hPSCs) have immense potential as a source of intestines. Therefore, an efficient system is needed for visualizing the stage of intestinal differentiation and further identifying hIOs derived from hPSCs. Here, 2 fluorescent biosensors were developed based on human-induced pluripotent stem cell (hiPSC) lines that stably expressed fluorescent reporters driven by intestine-specific gene promoters (KLF5mCherry and ISXeGFP). Then hIOs were efficiently induced from those transgenic hiPSC lines in which monomeric Cherry– or enhanced green fluorescent protein–expressing cells, which appeared during differentiation, could be identified in intact living cells in real time. Reporter gene expression had no adverse effects on differentiation into hIOs and proliferation. After orthotopic transplantation, the localization of the hIOs in the small intestine could be accurately visualized using FLI. Our study establishes a selective system for monitoring the in vitro differentiation and for tracking the in vivo localization of hIOs and contributes to further improvement of cell-based therapies and preclinical screenings in the intestinal field.
Keywords: KLF5, ISX, fluorescence imaging, reporter system, human intestine
Abbreviations: 3D, 3-dimensional; BMP, bone morphogenetic protein; DE, definitive endoderm; eGFP, enhanced green fluorescent protein; FLI, fluorescence imaging; hESC, human embryonic stem cell; HG, hindgut; hIO, human intestinal organoid; hiPSC, human-induced pluripotent stem cell; hPSC, human pluripotent stem cells; ISX, intestine-specific homeobox transcription factor; KLF5, Krüppel-like factor 5; mCherry, monomeric Cherry; NSG, NOD-SCID IL-2Rgnull; qRT-PCR, quantitative RT-PCR; SI, small intestine
INTRODUCTION
The small intestine (SI) is a multistructural and multifunctional organ that participates in the uptake of nutrients, provides a barrier function, modulates the intestinal microbiome, and provides host protection against harmful pathogens and toxic substances through a defense mechanism. The SI is lined with a complex epithelium that harbors region-specific architecture and multiple cell types comprising absorptive enterocytes and 3 types of secretory cells (Paneth, enteroendocrine, and goblet cells). In addition, to perform its appropriate functions, other specialized cells are integrated into the intestine, such as vascular, neuronal, lymphatic immune, and smooth muscle cells and fibroblasts. For several decades, studies in model organisms have substantially advanced our understanding of intestinal development and function. However, animal models have relatively low throughput and do not accurately mimic human physiology. Therefore, an in vitro human intestinal model represents a valuable platform for studying human intestinal physiology and disease.
Recently, alternative approaches have been developed to provide in vitro models that mimic human intestine tissues. Human intestinal organoids (hIOs) can be derived from human tissue samples, for example, from intestinal tissue biopsies from patients or from human pluripotent stem cells (hPSCs), which include human embryonic stem cells (hESCs) and human-induced pluripotent stem cells (hiPSCs), using directed differentiation. hIOs are three-dimensional (3D), multicellular structures that recapitulate aspects of the structural and functional features of the human SI. Several studies have shown that hPSC-derived hIOs contain the major cell types of the SI, including the diverse mesenchymal cells, such as smooth muscle cells, myofibroblasts, and fibroblasts, as found in the submucosal layer of the human SI. Moreover, hPSC-derived hIOs are capable of performing some physiologic functions, such as the absorption of dipeptides and secretion of protective mucus. Additionally, hPSC-derived hIOs can be passaged multiple times in vitro. These hIOs have been used to study human intestinal development and diseases and will be increasingly applied to many aspects of therapeutic treatment, such as transplantation and personalized medicine.
For both the study and use of hIOs in prospective applications, reliable methods for the identification, selection, expansion, and characterization of intestine-specific cell types in hIOs would be helpful. Intestine-specific reporter cell lines provide the opportunity to monitor cell-subtype specifications, to identify the molecular changes during in vitro differentiation into specialized cell types, and to understand the mechanisms of action of a variety of intestinal cells, both in vivo and in vitro, by tracking intestine lineage-specific cells. Fluorescent labeling of these cells is useful for evaluating the differentiation status of hPSCs toward intestinal cells and the distribution after transplantation into animal models. The data generated from such studies can facilitate the analysis of the differentiation status of hPSCs and the optimization and validation of protocols for intestinal differentiation of hPSCs. Furthermore, the development of sensitive and noninvasive methods for monitoring cell engraftment in vivo can translate from basic research to future clinical applications.
In the quiet contemplation of our surroundings, we often find that the most profound insights come from the harmony between nature and human well-being. Our appreciation for the natural world is deepened by understanding the History Of Blackberry Plants & Zoldonrasib Research – Approved Free Article, which reminds us of the resilience found in our environment. This holistic view extends to the vital importance of mental health, as discussed in the Jimma-Minnesota International Symposium on Mental Health and Substance Use, fostering a stronger global community. Furthermore, we value the scientific pursuit of nurturing life at its very beginning, highlighted by the Bafilomycin A1 – Membership Benefits – International Society for Research in Human Milk and Lactation, reflecting our shared commitment to a healthier and more enlightened future.
In this study, we generated genetically engineered hiPSC lines that allow for the identification and tracking of differentiating intestinal cells. This approach is based on the use of intestine lineage-specific promoters, such as the Krüppel-like factor 5 (KLF5), also known as intestine-enriched Krüppel-like factor and ISX, an intestine-specific homeobox transcription factor, to direct the expression of a reporter gene and selection marker. We used monomeric Cherry (mCherry) and enhanced green fluorescent protein (eGFP) as intestinal biosensors, such as KLF5mCherry or ISXeGFP reporter hiPSC lines; investigated the in vitro differentiation potentiality to improve the hIO differentiation protocols for hiPSC-derived hIOs; and performed in vivo transplantation experiments to monitor hIOs in 3D structures in real time. The results from this study can provide an efficient and selective fluorescent reporter system for the derivation of intestine lineage cells from hiPSCs and offer an approach for studying the clinical application of cell-based transplantation therapy to ensure the success of cell delivery by visualizing engrafted hIOs.
MATERIALS AND METHODS
Generation and Culture of hiPSC
hiPSCs were created from healthy human fibroblasts (CRL-2097; American Type Culture Collection, Manassas, VA, USA) with a nonintegrative reprogramming method and nonintegrating oriP/EBNA-1–based episomal vectors (A14703; Thermo Fisher Scientific, Waltham, MA, USA), as previously described. After electroporation, fibroblasts were plated at 1 × 10^5/well on Matrigel-coated, 6-well plates (BD Biosciences, San Diego, CA, USA) with mTeSR1 medium (StemCell Technologies, Vancouver, BC, Canada). After 2–3 weeks, hESC-like hiPSC colonies were selected and subsequently expanded for further characterization. Fibroblasts and hiPSCs were cultured as previously described.
Establishment of Stable KLF5mCherry and ISXeGFP Reporter hiPSC Lines
Stable KLF5mCherry and ISXeGFP reporter hiPSC lines were generated using lentiviral vectors containing the KLF5 promoter and mCherry reporter gene (HPRM23214-LvPM02; GeneCopoeia, Rockville, MD, USA) and the ISX promoter and eGFP reporter gene (HPRM12412-LvPF02; GeneCopoeia). Lentiviral particles were prepared as previously described, and transduction of the reporter constructs was performed by incubating recipient hiPSCs in culture medium supplemented with 8 mg/ml polybrene using a multiplicity of infection of 0.5. After selecting stable clones with puromycin (0.5 mg/ml; Sigma-Aldrich, St. Louis, MO, USA), we confirmed that the reporter system was functional by inducing intestinal differentiation and monitoring the reporter gene fluorescence with fluorescence microscopy (IX51; Olympus, Tokyo, Japan).
Differentiation of hiPSCs into hIOs
hIOs were generated as previously described with some modifications. To produce a definitive endoderm, undifferentiated hiPSCs were treated with 100 ng/ml Activin A (R&D Systems, Minneapolis, MN, USA) for 3 days with increasing concentrations of 0, 0.2, and 2% defined fetal bovine serum (GE Healthcare Life Sciences, Little Chalfont, United Kingdom). To differentiate into 3D aggregates called hindgut (HG) spheroids, cells were treated with 500 ng/ml fibroblast growth factor 4 (R&D Systems) and 500 ng/ml WNT3A (R&D Systems) for 4 days. Hindgut spheroids were embedded in Matrigel, cultured in hIO medium containing 1 times B27 (Thermo Fisher Scientific), 100 ng/ml epidermal growth factor (R&D Systems), 500 ng/ml R-Spondin 1 (R&D Systems) in the presence of 100 ng/ml Noggin (R&D Systems), and 0.5 mM DMH1 (Bio-Techne, Minneapolis, MN, USA) or 50 nM LDN-193189 (Sigma-Aldrich), 2 novel highly selective bone morphogenetic protein (BMP) inhibitors, and then passaged every 2 weeks.
Real-Time Quantitative RT-PCR
Total RNA was extracted from cells with an RNeasy Kit (Qiagen, Valencia, CA, USA) and reverse-transcribed using a Superscript IV First-Strand Synthesis System Kit (Thermo Fisher Scientific) as described previously. Real-time quantitative RT-PCR (qRT-PCR) was performed using Power SYBR green PCR Master Mix (Thermo Fisher Scientific) on a 7500 Fast real-time PCR system (Thermo Fisher Scientific), as previously described.
Immunocytochemistry and In Vitro Fluorescence Imaging
Immunocytochemistry was performed as previously described. In brief, cells were fixed in 4% paraformaldehyde and then permeabilized with 0.1% Triton X-100. hIOs and tissue samples were fixed and frozen in optimal cutting temperature compound (Sakura Finetek Japan, Tokyo, Japan). Frozen samples were cut at 10–20 μm using a cryostat microtome at −20°C and permeabilized with 0.1% Triton X-100. After being blocked with 4% bovine serum albumin, samples were incubated with specific primary antibodies at 4°C overnight, followed by incubation with the corresponding Alexa Fluor 488–, -594–, or -647–conjugated secondary antibodies for 1 hour at room temperature. Paraffin sections were deparaffinized, which was followed by antigen retrieval in citrate buffer and staining in a manner similar to optimal cutting temperature sections. The nuclei were stained with DAPI (1 mg/ml; Thermo Fisher Scientific). Samples were observed with a fluorescence microscope (IX51; Olympus) and an Axiovert 200M microscope (Carl Zeiss, Oberkochen, Germany). The number of each type of marker-positive cell per field of vision was quantified in ≥3 fields of vision per condition using ImageJ software (National Institutes of Health, Bethesda, MD, USA) and is presented as the percentage of the total number of cells counted.
Transplantation
NOD-SCID IL-2Rγnull (NSG) mice (The Jackson Laboratory, Bar Harbor, ME, USA) were housed in a standard animal-maintenance facility under a 12:12-h light:dark cycle at a constant temperature (20–22°C). All experiments were performed after approval by the institutional animal care and use committee of the Korea Research Institute of Bioscience and Biotechnology (approval KRIBB-AEC-16206). The procedure for the kidney capsule xenografting of hIOs was performed as described previously. A right subcostal incision was made to expose the kidney. hIOs were transplanted into the subcapsular space of the kidney, and the skin was closed in a double layer. The mice were kept warm with a heating pad until they fully recovered from anesthesia. At 1 week after transplantation, the mice were humanely euthanized, and xenografts were isolated for analysis. The SI xenografting of hIOs was performed as previously described. Chopped hIO fragments were resuspended in 100 ml of diluted Matrigel in PBS (1:20) and instilled into the intestinal lumen using a syringe. After intraintestinal infusion, the anal verge was sutured for 6 hours. Animals were subsequently euthanized at 3 hours and at 7 days.
Ex Vivo Fluorescence Imaging
To determine the transplantation of hIOs, in vivo fluorescence imaging (FLI) was performed on the InVivo Imaging Systems Lumina II (Xenogen, Alameda, CA, USA). Fresh organs (SI, heart, lung, liver, and kidney) were analyzed for fluorescence using the IVIS system. eGFP was excited at 465 nm and detected at 509 nm, and mCherry was excited at 535 nm and detected at 592 nm. The fluorescence images were shown in radiance photons, and the epifluorescence intensity represented the average radiance [photons/second/cm²/steradian (p/s/cm²/sr)].
Histology and Live-Cell Imaging
Imaging of tissue samples after hIO transplantation was performed with a fluorescence microscope (IX51; Olympus). SI structures were observed using brightfield and differential interference contrast microscopy, and xenografts of hIOs were imaged with a fluorescence microscope. Live-cell imaging of hIOs was performed on a motorized Olympus IX83 inverted microscope with an on-stage incubator (Live Cell Instrument, Seoul, South Korea). Time-lapse images were acquired every 1 hour for 48 hours, and processing was performed using ImageJ software.
Short Tandem Repeat and Karyotype Analysis
Short tandem repeat analysis of human fibroblasts and the hiPSC line was performed by Humanpass (Seoul, South Korea). Karyotype analysis using the standard chromosome G-banding method was conducted by GenDix, (Seoul, South Korea).
In Vitro Differentiation Through the Formation of Embryoid Bodies
For spontaneous differentiation into the 3 germ layers, hiPSCs were dissociated by treatment with 1 mg/ml collagenase IV and plated onto petri dishes in embryoid body medium, consisting of knockout DMEM (Thermo Fisher Scientific) supplemented with 10% knockout serum replacement (Thermo Fisher Scientific), 1% nonessential amino acids (Thermo Fisher Scientific), 0.1 mM β-mercaptoethanol (Sigma-Aldrich), and 1 mM L-glutamine (Thermo Fisher Scientific). After 5 days in suspension culture, embryoid bodies were attached to gelatin-coated LabTek chamber slides (Nalge Nunc, Rochester, NY, USA) and cultured for an additional 1 week.
Teratoma
hiPSCs were subcutaneously injected into 6-week-old, specific pathogen-free/viral antibody-free immunodeficient mice (Orient Bio, Gyeonggi, South Korea). After 10–12 weeks, the teratomas were harvested, fixed in 4% paraformaldehyde, embedded in paraffin, and stained with hematoxylin and eosin solution (Sigma-Aldrich). Animal experiments were approved by the Institutional Animal Care and Use Committee of Korea Research Institute of Bioscience and Biotechnology (approval KRIBB-AEC-17014).
RESULTS
KLF5 and ISX Expression Increased During Intestinal Differentiation
Gene expression data from the human protein atlas revealed that the transcription factor KLF5 was found in the various tissues and organs examined in this study, with particularly high levels in the digestive organs, whereas the intestine-specific transcription factor ISX was exclusively expressed in the intestinal tract, including the duodenum, SI, colon, and rectum. Similar results were also observed in a meta-analysis of 38 different microarray data sets from undifferentiated hPSCs, organ-specific tissues, and various cell types. Real-time qRT-PCR analysis also confirmed that KLF5 was relatively highly expressed in the liver, lung, stomach, SI, and colon, while ISX was detected only in the intestinal tissues.
Next, we sought to determine whether expression of KLF5 and ISX increased during directed differentiation of hPSCs into intestinal tissue in vitro. Fully characterized hiPSCs were used for intestinal differentiation. Using the previously described differentiation protocol for a 3D, intestinal, tissue-like structure, hiPSCs were efficiently differentiated into a definitive endoderm (DE), HG, and hIO, as indicated by their distinct morphologies. A progressive increase in the expression of both KLF5 and ISX was observed during intestinal-directed differentiation. Notably, KLF5 expression increased before the increase in ISX. The expression of KLF5 was induced during the entire period of differentiation, particularly from the DE stage, whereas the expression of ISX was up-regulated ~40 days after the start of the differentiation process (hIO p2). These results indicated that KLF5 is primarily expressed in digestive tissues but is also expressed in various tissues and is expressed from the early stage of intestinal differentiation, whereas ISX is specifically expressed in intestinal cells and the tissues of the adult human SI and colon. Therefore, we hypothesized that the early KLF5-expressing, intestinal-lineage commitment cells and the specialized ISX-expressing intestinal epithelial cells could be identified and visualized with KLF5 and ISX reporter constructs.
KLF5mCherry and ISXeGFP Biosensors Can Monitor the Process of Intestinal Differentiation
We developed an intestinal biosensor cell line via the stable transfection of hiPSCs with either the lentiviral KLF5mCherry or ISXeGFP promoter reporter. To demonstrate that the KLF5mCherry hiPSC line and ISXeGFP hiPSC line faithfully recapitulated ISX and KLF5 promoter activity, respectively, hiPSCs were differentiated into hIOs using the stepwise differentiation protocol. The red fluorescence of mCherry was clearly detectable in a subset of cells 3 days after intestinal differentiation (HG stage). Cells expressing eGFP appeared in a late stage of differentiation, after hIOs had formed completely. Remarkably, the more that hIOs acquired differentiated phenotypes after a series of passages (p2), the greater was the expression of eGFP, which was pronounced in the epithelium of hIOs, compared with mCherry expression. Real-time FLI showed that the number of both eGFP- and mCherry-expressing cells increased as hIOs matured and proliferated, consistent with the previous observation that ISX expression is limited to the epithelial lining of the fetal and adult intestine.
We assessed the expression and localization of intestinal transcription factors, including SOX9, CDX2, ISX, and KLF5, and intestinal cell markers, including VIL (villin 1 for enterocytes), CHGA (chromogranin A for enteroendocrine cells), LYZ (lysozyme for Paneth cells), and MUC2 (mucin 2 for goblet cells), in hIOs with marker-specific antibodies, showing that hIOs contained all intestinal epithelial cell types. The ratios of each type of marker-positive cell were not significantly different in the differentiated hIO samples. As expected, ISX promoter-driven eGFP was exclusively expressed in hIOs derived from the ISXeGFP hiPSC line (hIOs-ISXeGFP), whereas KLF5 promoter-driven mCherry was selectively expressed in hIOs derived from the KLF5mCherry hiPSC line (hIO-KLF5mCherry). The differentiated hIOs expressed mRNA transcripts for specific markers of intestinal transcription factors, including KLF5, ISX, CDX2, and SOX9, and 4 major cell types in the SI, including LYZ, VIL1, MUC2, and CHGA; however, their levels were lower than those in the human SI control, as previously reported. These results suggested that there was no observable difference in the intestinal differentiation potential between control and reporter hiPSC lines.
KLF5mCherry and ISXeGFP Biosensors Provide a Rapid and Efficient Screening Method for Intestinal Differentiation-Inducing Compounds
Next, we tested whether the reporter system described here could be used to assess intestinal differentiation efficiency. We examined the effect of several chemical inhibitors of BMP signaling on the differentiation of hIOs and concluded that DMH1, a highly selective small-molecule inhibitor of BMP, is an effective and efficient supplement for hIO differentiation and maintenance. To improve differentiation efficiency, we modified the hIO medium by replacing the recombinant Noggin peptide (an antagonist of BMP-4) with DMH1 using KLF5mCherry hiPSC and ISXeGFP hiPSC lines. DMH1 led to a significant increase in the size of hIOs as well as in the number of budding structures with an accompanying significant increase in the fluorescence intensity of the hIOs. Even at the early stage of intestinal differentiation (at hIO p1), the fluorescence of mCherry and eGFP was clearly more visible in hIOs-KLF5mCherry and hIOs-ISXeGFP cells, respectively, when using our optimized protocol with DMH1. Most important, the greater level of fluorescence in hIOs treated with DMH1 resulted in an overall increase in the mRNA levels for markers of intestinal differentiation, showing that there was a strong correlation between the differentiation efficiency and the fluorescence signal. These results demonstrated that both the KLF5mCherry and ISXeGFP promoter reporters allowed for the visualization of KLF5- and ISX-expressing intestinal cells during hiPSC differentiation into hIOs.
Ex Vivo Visualization of the Grafted hIOs
hIOs-ISXeGFP, which was the optimal promoter system for following intestine-specific gene expression, stably expressed the eGFP fluorescence in a dose-dependent manner, as demonstrated by a positive linear correlation (R² = 0.958) between the number of hIOs and the fluorescence intensity. Given that there was a linear relationship between the number of hIOs and the fluorescence intensity, that imaging approach will enable us to noninvasively track and quantify the survival and growth of the grafted hIOs. For quantitative detection of the transplanted hIOs, the hIOs-ISXeGFP were implanted under the kidney capsule of immunodeficient NSG mice for imaging of the xenografts. A clear fluorescent signal was observed in the kidney 1 week after transplantation when the kidneys were removed. The IVIS imaging system showed that the fluorescence intensity of 7 hIOs-ISXeGFP produced in vitro was increased in the same number of transplanted hIOs-ISXeGFP [average radiance: 4.023 (p/s/cm²/sr) × 10⁷ and average radiance: 4.536 (p/s/cm²/sr) × 10⁸]. These data indicate that the fluorescent signal was maintained during in vivo transplantation and was further enhanced throughout the growth of the hIOs-ISXeGFP in vivo.
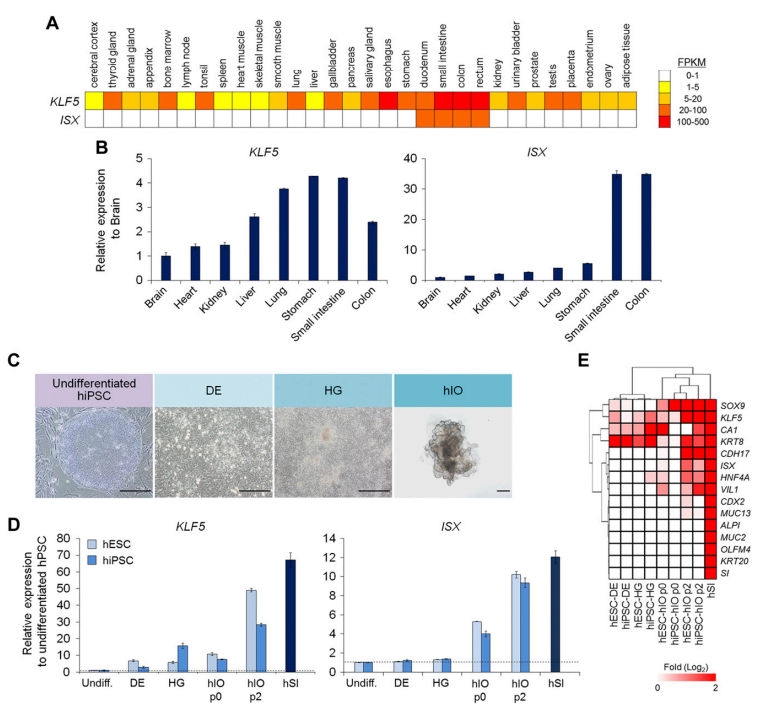
Figure 1. Intestine-Specific Expression of KLF5 and ISX.Gene transcriptional levels in 30 tissues from the Human Protein Atlas are displayed using the values of 4 different abundance levels for fragments per kilobase of transcripts, per million fragments sequenced (FPKMs): 1–5 FPKMs (yellow), 5-20 FPKM (orange), 20-100 FPKM (light red), and 100-500 FPKM (red). Quantitative PCR analysis of KLF5 and ISX expression in various tissues. Fold changes in the expression levels are relative to the expression in the brain. Representative cell morphology during the hIO differentiation process. Scale bars, 500 µm. Real-time qRT-PCR analysis of KLF5 and ISX expression in undifferentiated (Undiff.) hPSCs (H9 hESCs and hiPSCs derived from human fibroblasts), DEs, HGs, differentiated hIO (p0, p2), and human small intestine (hSI). Heatmaps of the microarray expression data for the intestinal markers.
The histologic examination revealed that the hIOs-ISXeGFP and hIOs-KLF5mCherry successfully engrafted under the kidney capsule of the recipient mice. The transplanted hIOs were shown as small, closed spheres with luminal structures. The engrafted hIOs-ISXeGFP and hIOs-KLF5mCherry in vivo were eGFP+ and mCherry+, respectively. The presence of intestinal cell types and the human phenotype of the grafted eGFP-expressing or mCherry-expressing cells were also verified by costaining for human intestine-specific antibodies. These intestine-specific, marker-expressing cells were detected in the epithelium of transplanted hIOs. Ki-67 expression in transplanted hIOs has been shown to indicate their proliferative capacity. Given that the ex vivo eGFP and mCherry fluorescent signals showed the distribution of the transplanted hIOs, we suggest that the promoter–reporter biosensor-expressing hIOs may provide an opportunity to track the localization and engraftment of hIOs after transplantation.
Engraftment of the hIOs-KLF5mCherry and hIOs-ISXeGFP in the SI
To investigate the availability of our intestine-specific biosensor hIOs in a more physiologically relevant context, we transplanted hIOs-KLF5mCherry and hIOs-ISXeGFP into the intestinal lumen of the SI, which is the ideal target site for hIO transplantation. Within 3 hours after transplantation, the SI tissues were imaged for fluorescence using the IVIS imaging system. A fluorescent signal was detected in the SI of mice transplanted with hIOs-KLF5mCherry and hIOs-ISXeGFP with an average radiance of 3.06 ± 0.67 (p/s/cm²/sr) × 10⁸ and 2.35 ± 0.12 (p/s/cm²/sr) × 10⁸, respectively. The localization of the engrafted hIO-ISXeGFP or hIO-KLF5mCherry was directly visualized using fluorescence microscopy. Most important, even 1 week after transplantation, the hIO-KLF5mCherry- and hIO-ISXeGFP-engrafted SI exhibited significantly greater fluorescence intensity (8.12 ± 1.06 (p/s/cm²/sr) × 10⁸ and 8.25 ± 3.15 (p/s/cm²/sr) × 10⁸ in the hIOs-KLF5mCherry- and hIOs-ISXeGFP-engrafted SI, respectively) than the average radiance in nonfluorescent organs, such as the heart, lung, liver, and kidney (P < 0.01 in the hIO-KLF5mCherry-engrafted SI and P < 0.05 in the hIO-ISXeGFP-engrafted SI). These data raised the possibility that our intestine-specific biosensor system could be useful for studying intestinal transplantation.
DISCUSSION
Real-time imaging of hPSC fate has made a significant contribution to the successful application of these cells in various aspects of translational applications in vitro and in vivo. In this article, we describe the establishment of transgenic hiPSC lines that allow for the identification and selection of differentiating human intestinal cells via hIO models. This approach is based on using intestine-specific promoters to drive the expression of a selectable reporter gene. Our results demonstrate that, during the in vitro differentiation of the transgenic hiPSC lines, the eGFP- or mCherry-expressing cells could be identified. These fluorescence-expressing cells were stained positively for intestine-specific proteins, expressed intestine-specific genes, and could form stable intestinal grafts after in vivo hIO transplantation. The fluorescence intensity of hIOs derived from the transgenic hiPSC lines was correlated to the number of intestinal cells.
Reporter lines using tissue-specific promoters for specific cell lineages have been described previously in hPSCs and have allowed the identification and selection of cardiomyocytes, hepatocytes, neurons, and pancreatic β cells. Although the leucine-rich, repeat-containing G-protein coupled receptor 5 (LGR5)-GFP transgenic reporter hESC line using a bacterial artificial chromosome or zinc-finger nuclease was generated and used to trace only intestinal stem cells, to our knowledge, this is the first report describing fluorescent hiPSC reporter lines that detect intestinal epithelial cells and have a potential use in the development and optimization of efficient intestinal differentiation protocols. We chose to use the ISX and KLF5 promoters because we noted robust expression of those genes in hIOs generated during the in vitro differentiation of hPSCs in our previous study using microarrays. Previous studies have also shown that KLF5 is present in the proliferating cells of various digestive tissues, even in the esophagus, whereas ISX is expressed only in the epithelia of the intestine. We observed that the expression of eGFP was more intense than that of mCherry in the epithelium of hIOs. Most important, we used these intestinal promoter-driven fluorescence reporters in an assay for phenotypic differentiation of hIOs and found that DMH1 (a BMP pathway inhibitor) can induce intestinal differentiation by substituting for Noggin in live cells. The hIO differentiation efficiency was determined rapidly and simply using our reporter system. In addition, the generation of the transgenic hiPSC lines did not have any negative effect on their ability to differentiate into 3D hIOs comprising all intestinal epithelial cell types with their specific structure when compared to that of the wild-type lines. Because hIOs contain multiple cell types, a detailed strategy for fine-tuning the hIOs-KLF5mCherry and hIOs-ISXeGFP models is needed to identify and characterize subpopulations of the various cell types in the hIOs.
Intestinal transplantation can be a curative treatment for patients with intestinal disease, including short bowel syndrome, enterocolitis, Crohn’s disease, intestinal motility disorders, and rare genetic intestinal diseases, such as Hirschsprung’s disease. However, the success rate of intestinal transplantation is very low because of the frequent failure of the initial intestinal cell engraftment and the high risk of rejection because of the complex nature of the SI immune system. Therefore, noninvasive imaging of transplanted intestinal cells is necessary for understanding their interactions with host environments in vivo, for tracking cell fate and function, and for monitoring patient safety. In this study, we used an FLI system to visualize the fate of grafted, hiPSC-derived hIOs under the kidney capsule or the SI lumen of immunodeficient NSG mice. Because hIOs had intestinal biosensors containing promoter–reporter constructs (KLF5mCherry or ISXeGFP), the transplanted hIOs were tracked with FLI. hiPSC-derived hIOs offer the possibility of modeling intestinal disease in a patient-specific manner and providing a cell source for autologous transplantation of the intestine. Therefore, these fluorescent biosensors for hIOs could be applied in cell-based transplantation studies for the visualization of transplanted intestinal cells and the study of the functional integration and maturation of the cells into the host intestine. In addition to the in vivo approaches, these reporter-based strategies have a potential role for the development and optimization of hIO-based in vitro assays for drug discovery and disease modeling.
In conclusion, this report describes the generation of fluorescent reporter systems that allow for the identification and tracking of hiPSC-derived hIOs that contain crypt and villus-like structures and all 4 major cell types, recapitulating the cellular diversity and architecture of the intestinal epithelium. Our KLF5mCherry or ISXeGFP reporter system could serve as an ideal biosensor of intestinal fate and differentiation in live cells. This ability may have important implications for several intestinal research fields, including development research, physiologic and pharmacologic studies, tissue engineering, and therapeutic transplantation.
