Rapamycin, Cyclosporin A, and Tacrolimus (FK506): A Comparative Review of Transplant Immunosuppressants and FKBP Inhibition
Immunosuppressive drugs used in the transplantation period are generally defined as induction and maintenance therapy. The use of immunosuppressants, which are particularly useful and have fewer side effects, decreased both mortality and morbidity. Many drugs such as steroids, calcineurin inhibitors (cyclosporine-A, tacrolimus), antimetabolites (mycophenolate mofetil, azathioprine), and mTOR inhibitors (Rapamycin, sirolimus, everolimus) are used as immunosuppressive agents.The integration of Rapamycin and its derivatives has allowed for “calcineurin-sparing” protocols, which help mitigate long-term nephrotoxicity—a significant advancement in extending the lifespan of transplanted organs.
Although immunosuppressant drugs cause many side effects such as hypertension, infection, and hyperlipidemia, they are the agents that should be used to prevent organ rejection. This shows the importance of individualized drug use. The optimal immunosuppressive therapy post-transplant is not established. Therefore, discovering less toxic but more potent new agents is of great importance, and new experimental and clinical studies are needed in this regard. Our review discussed the mechanism of immunosuppressants, new agents’ discovery, and current therapeutic protocols in the transplantation.
KEYWORDS
Immunosuppressant; transplant pharmacist; signaling pathways; rejection; molecular biology
Introduction
Long-term patients and graft survival increased dramatically with solid organ transplantation forms due to developments in immunosuppressants. Developments in immunosuppressants that avoid acute reactions and success in transplantation surgery are among the critical reasons for this Increase in sustained graft survival. Acute graft rejection and chronic allograft disease remain a big concern in today’s transplantation surgery, where advanced immunosuppressive procedures are used. Long-term application of immunosuppressive therapy results in a variety of adverse effects and toxicity . While the multi-drug solution reduces toxicity by reducing each drug’s target doses, it is a fact that this technique greatly impacts the quality of life of patients.
Immunosuppressive agents are known as induction and maintenance treatment. The purpose of the treatment is to prevent the rejection of graft and to provide induction of tolerance. While several medications are used because of these treatment protocols, there is still no perfect immunosuppressive agent. In modern days, the individualization of immunosuppressive regimens is possible by incorporating new agents into the protocols. The timeline for transplant related immunosuppressive drugs is shown in Figure 1.
Transplant immunology
The immune system is the primary barrier to long-term graft survival in solid organ transplant recipients. The donor organ may be rejected by the recipient’s immune system as a foreign substance. The ultimate goal of transplant recipients is graft acceptance, known as tolerance, ideally without longterm immunosuppressant drugs.
Major histocompatibility complex/human leukocyte antigens molecules
Major histocompatibility complex (MHC) genes encode the most potent transplant antigens. In humans, these MHC molecules are called human leukocyte antigens (HLA). It is divided into Class I molecules and Class II molecules. Class I molecules are expressed on all nucleated cells and usually present endogenous minor antigens such as viruses and self-proteins fragments in the context of self-MHC to CD8+ T. Class II molecules are constitutively expressed only on professional antigen-presenting cells (APC), including dendritic cells, macrophages, and B cells. After exposure to proinflammatory cytokines, their expression can be upregulated in epithelial and vascular endothelial cells. Class II molecules present relatively more significant antigens derived from extracellular proteins to CD4+ T-cells . The degree of HLA mismatch between donor and recipient plays a role in determining the risk of chronic rejection and graft loss. HLA-A, -B, and -DR (3 pairs, 6 antigens) are traditionally used for typing and matching before kidney or pancreas transplantation. HLA-Cw, -DP, and DQ are now increasingly used in many transplant centers. For kidney transplants, long-term graft survival is best in HLA-identical living-associated kidney transplants. The most significant effect comes from the coupling of the DR antigen, and the order of importance for HLA matching in kidney transplantation is DR > B > A.
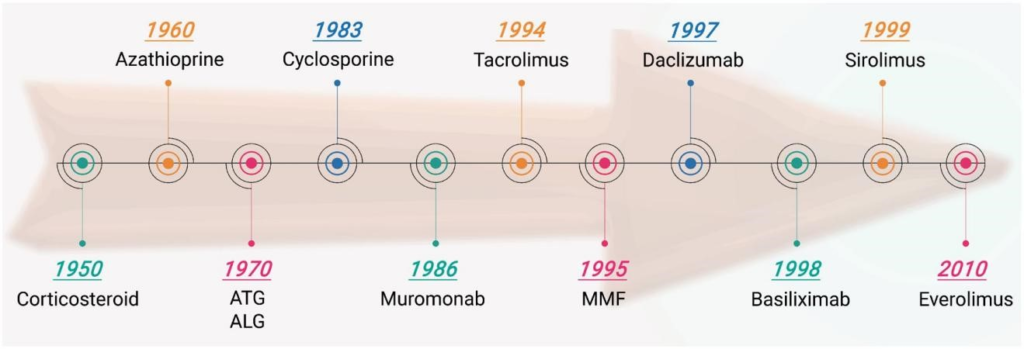
Figure 1. Timeline of transplant-related immunosuppressant drugs.
Non-HLA antigens/antibodies
Acute and chronic graft rejection can occur in HLA-identical sibling transplants, indicating the presence of an immune response to non-HLA antigens. There are several non-HLA antigens, and their antibodies derived from either alloimmunity or autoimmunity have been reported.
ABO blood group antigens
ABO blood group antigens are expressed not only on red blood cells but also on other cells. ABO-incompatible organ transplants cause hyperacute rejection due to the presence of preformed hemagglutinin A and B antibodies. ABO compatibility between donor and recipient is required for organ transplantation. Desensitization protocols have been used for ABO-incompatible kidney transplants . Rhesus factor and other red cell antigens are not relevant for organ transplantation as they are not expressed in the endothelium.
Minor histocompatibility antigens (MiHA)
MiHA are small endogenous peptides that occupy the antigen-binding site of donor MHC molecules. They are usually recognized by CD8+ cytotoxic T-cells, leading to graft rejection. MiHA plays an essential role in graft-versus-host disease in patients who receive HLA-matched cells in bone marrow transplantation. MHC class I-related chain A and B (MICA and MICB) are also expressed in endothelial cells. Antibodies to MICA and MICB can cause antibody-mediated rejection and graft loss.
Three-signal model of t-cell activation
Awareness of immunosuppressive agents’ immunological reaction to solid organ transplantation tolerance is essential . The adaptive immune response to the donor grafting starts with identifying alloantigen by a pure T cell. And then, the T cell starts to proliferate and to differentiate . This main event’s frequency depends on the association between the T cell receptor (TCR) and the antigen provided as peptide by the APC and the receptor/ligand interaction. The active T cells have cytotoxic effects. It can interact with the B cell for antibody production and cause hypersensitivity to the macrophages . The three-signal T cell activation and proliferation model offer details on the molecular mechanism and domain of immunosuppressive drugs .
Signal 1 (alloantigen recognition)
Signal 1 includes APCs (macrophages and dendritic cells) presenting the foreign antigen to T lymphocyte. It activates the TCR, which enables signal transduction through the transduction apparatus known as the CD3 complex.
Signal 2 (lymphocyte activation, co-stimulation) Signal 2 is an antigen nonspecific co-stimulatory signal generated due to the process in which several ligands on the APC bind to various TCRs, including CD28, CD154, CD2, CD11a, and CD54. Both signal 1 and signal 2 allow activation of the calcium–calcineurin pathway, the mitogen-activated protein (MAP) pathway, and the nuclear factor-jB (NF-jB) pathway. Calcineurin inhibitors can inhibit this signaling mechanism.
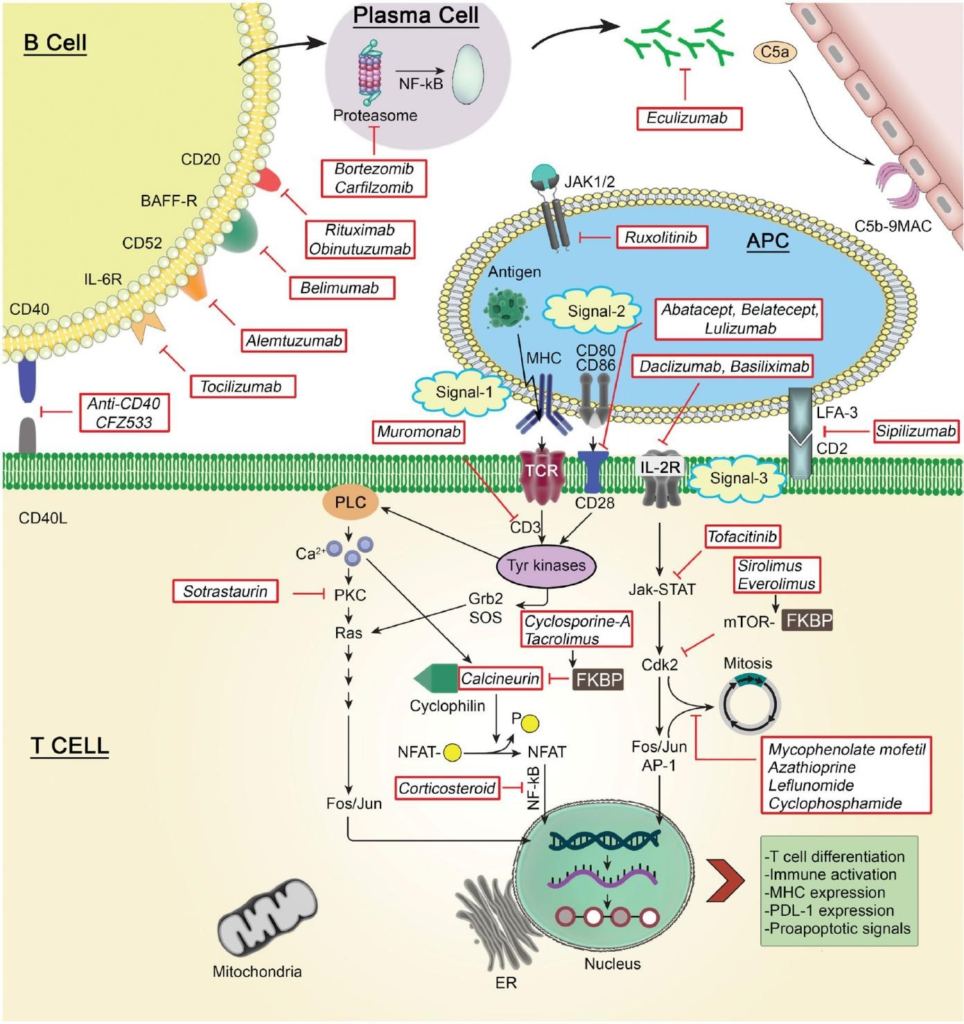
Figure 2. Mechanisms of action and targets of transplant-related immunosuppressant drugs.
Signal 3 (clonal expansion)
As a result of this interaction, the expression of IL-2 is increased, which stimulates the cell cycle through the receptor (IL-2R). Signal 3 requires the enzyme target of Rapamycin mTOR inhibitor benefits for activation, mRNA translation, and cell proliferation. Monoclonal antibodies can inhibit this signaling pathway against the IL-2 receptor, mTOR inhibitors, and antiproliferative agents. Immunosuppressive agents can mainly affect cytokines’ release/production by activated T cells, inhibit T cell proliferation and TCR, or cause T cell depletion . The mechanisms of action and targets of transplant-related immunosuppressant drugs are shown in Figure 2.
Novel immunosuppressive agents
Belatacept
Belatacept is a specific T cell activation blocking agent. It consists of the recombinant extracellular region of human cytotoxic T lymphocyte antigen-4 (CTLA-4) and a portion of the modified Fc portion of human IgG-1. The agent binds to CD 80/86 ligands of antigen stimulating cells to inhibit CD28 mediated T cell activation . T cell activation and differentiation depend on the strength of the signal received by the TCR and the signals provided by co-stimulatory molecules . CD80/86 ligands of the co-stimulatory molecule in the APCs must be bound to CD28 of the T cell to induce the immunological response . In addition, many studies highlighted the inhibitory roles of CD80/CD86 and PDL-1/PDL-2 (in APCs) and CTLA-4 and programmed death PD-1 (expressed in T cells), respectively . Belatacept can block this interaction and significantly prevent the development of rejection in kidney transplantation. In vitro studies have also been shown to prevent CD4 and CD8 T cell proliferation .
Belatacept’s mechanism of action is similar to abatacept; however, there is a small structural difference. Belatacept binds to CD86 with four times higher affinity than abatacept. It also binds to CD80 with an affinity two-fold higher than abatacept. While abatacept is used in treating dermatological and rheumatological diseases, belatacept is mostly used as a maintenance immunosuppressive drug in the postkidney transplant period, instead of calcineurin inhibitors. The half-life of belatacept is about 8–10 days and is administered as an IV infusion within 30 min. Advantage dose adjustment is not required in cases of liver and kidney failure . It has no interaction with other immunosuppressive drugs. Generally, in the first month after transplantation, a total of four doses are given at a dose of 10 mg/kg, the same dose in the next 3 months, and a dose of 5 mg/kg in the following months .
In the experimental kidney transplantation model performed in 1996, T cell co-stimulation blockade has been shown to prevent chronic rejection development. Also, in the experimental rat kidney transplant model, co-stimulation blockade has been shown to suppress the cellular and humoral immune response . Then BENEFIT study research was carried out. In this study, GFR rates were better in the belatacept group at the end of the 12th month, graft survival was comparable, but acute rejection rates and risk of posttransplant lymphoproliferative disease were higher in the belatacept group . Patients who used cyclosporine-A or Tacrolimus (FK506) FKBP inhibitor level were transferred to belatacept in another study, and these patients were followed-up. An improvement in GFR rates was reported at the end of the second year. As a result of these studies, the Food and Drug Administration (FDA) approved belatacept in 2011 for rejection prophylaxis in adult organ transplantation.
In a study on 666 patients who underwent kidney transplantation and prepared for 7 years, the belatacept’s efficacy and safety results were analyzed. According to the study, 7 years after transplantation, GFR was better in the belatacept group than in the cyclosporine-A group. The rate of death or graft loss was 43% less in the belatacept group. In terms of risk of infection development, CMV infection rates were similar in patients using belatacept and cyclosporine-A; however, the EBV reactivation rate is higher in the belatacept group.
Compared with the calcineurin inhibitor, belatacept can be used in both induction and maintenance treatment. It has more advantages in terms of chronic allograft nephropathy development, GFR protection, blood pressure control, hyperlipidemia, and diabetes development; it has been shown to have similar effects on acute rejection development and graft loss.
Belimumab
B-lymphocytes play an essential role in maintaining immune system function. B-lymphocyte stimulant (BLyS), or B cell-activating factor (BAFF, CD257), is a critical cytokine in the proliferation of B-cells . Belimumab is a human recombinant IgG1k monoclonal antibody that inhibits soluble BlyS . As a result, apoptosis occurs, circulating B cell clones decrease, conversion of B cells to plasma cells is blocked . Belimumab is ineffective for membrane-bound BAFF. As a dosage regimen, it is recommended as 10 mg/kg on the 0th, 14th, and 28th days and at four-week intervals. It should be given as an infusion in 1 h; however, the administration as a bolus is not recommended. Although it is not very clear, dose adjustment is not required in renal and liver failure. Hypersensitivity and infusion reactions (anaphylactic reactions, bradycardia, hypotension, angioedema, and dyspnea) can occur during or after administration. The incidence of these side effects is less than 1%.
In clinical practice, premedication (such as antihistamine, antipyretic) is recommended. Belimumab with other immunosuppressants, especially those targeting B lymphocytes, such as anti-CD20 therapies, may increase severe infection risk . Similarly, it is not recommended to combine belimumab with IV cyclophosphamide or live vaccines. Special attention should be paid to the development of progressive multifocal leukoencephalopathy in patients with systemic lupus erythematosus. Psychiatric influences, such as anxiety and depression, have also been reported. Live vaccines should not be administered to patients receiving belimumab. In a study in monkeys, belimumab caused a decrease in the number of B lymphocytes in the spleen and lymph nodes, depending on the dose . In a study of patients with high soluble BAFF levels, it was shown that the risk of developing DSA was higher, and this was also associated with the risk of developing graft dysfunction .
Preclinical and clinical research on the use of belimumab in solid organ transplantation is ongoing. A positive correlation was found between BAFF levels and human HLA I and HLA II antibodies in renal transplant patients . A multi-center, randomized, double-blind, placebo-controlled, phase II, a pilot study was performed comparing belimumab to placebo. According to the data obtained from this study, belimumab did not increase infection risk.
Bortezomib
Bortezomib is an anti-cancer medication used to treat multiple myeloma and mantle cell lymphoma . Also, its effectiveness in transplantation is investigated due to its immunosuppressant feature . Also, bortezomib causes plasma cell apoptosis. In vitro and in vivo (murine and human) studies have shown that this drug induces apoptosis of CD-138+ plasma cells.
Bortezomib also has effects on circulating B cells and TH cells. It can lead to blockade of the T-cell cycle, which leads to apoptosis of TH cells, and causes an IL-6 reduction. Peak plasma concentration of bortezomib is 60–120 ng/ mL following repeated doses of 1–1.3 mg/m2. The subsequent elimination half-life ranges from 40 to 190 h. While inhibitors and inducers of CYP3A4, CYP2C19, CPY1A2, and to a lesser extent, CYP2D6 and CYP2C9 are particularly common in organ transplant populations, clinically significant interactions with bortezomib and enzyme inducers are not very high.
This drug’s main adverse effect is dose-dependent peripheral sensory neuropathy, which occurs in about 30% of patients treated. Other side effects observed in patients receiving bortezomib treatment include nausea (55%), diarrhea (44%), thrombocytopenia (28%), neutropenia (11%), and fatigue (12%). Bortezomib may be positively associated with shingles, and prophylactic acyclovir may reduce the risk of this . Ocular side effects such as chalazion or hordeolum may be more common in women and cause discontinuation of therapy. Also, acute interstitial nephritis has been reported.
In a study in patients with relapsed or refractory multiple myeloma, bortezomib’s efficacy (with or without dexamethasone) was determined. In the phase III study, bortezomib was more effective than a high-dose dexamethasone regimen . Although studies on the selected patients are limited, however, based on data from kidney transplant recipients, this agent can be considered an alternative strategy against refractory antibody-mediated rejection.
Carfilzomib is a second-generation proteasome inhibitor that irreversibly binds to and inhibits the chymotrypsin-like activity of the 20S proteasome. One advantage of carfilzomib over bortezomib is the reduced risk of neuropathy. It may be part of a desensitization strategy in kidney transplant candidates. Studies have shown that desensitization based on carfilzomib monotherapy leads to significant bone marrow plasma cell depletion and anti-HLA antibody reduction while providing an acceptable safety and toxicity profile. As a result, both bortezomib and carfilzomib show their effects by inhibiting structural and immunoproteasomes. However, bortezomib has reversible activity, a shorter effective half-life, and selective proteasomal activity .
Janus kinase (JAK) inhibitors
JAKs are an important group of receptors that trigger the cytokine receptor through signal converter and activator of transcription (STAT) proteins . The JAK family consists of four members: JAK1, JAK2, JAK3, and tyrosine kinase 2 (Tyk2). Each cytokine receptor needs a pair of related two JAKs to signal through the cytokine receptor . JAK2 acts on the Type 1 erythropoietin cytokine receptor, whereas JAK3; is highly expressed on lymphocytes and other hemato-lymphoid cells. Once activated, it causes the STAT4 transcription factor’s dimerization and the stimulation of some pro-inflammatory cytokines such as IL-2, IL-4, IL-7, IL-9, IL-15, and IL-22. JAK inhibition inhibits many cytokine releases .
Tofacitinib
Tofacitinib is a low molecular weight drug with a potent immunomodulator and selective JAK enzyme inhibitor. Tofacitinib inhibits cytokine signaling through the IL-2Rp chain cytokine receptor and stimulates the apoptosis of T cells, B cells, and natural killer (NK) cells. Tofacitinib is a specific inhibitor of JAK3 (IC50 ¼ 2 nM), thus, blocking the activity of IL-2, IL-4, IL-15, and IL-21. Therefore, Th2 cell differentiation is inhibited, and, thus, tofa- citinib is useful in treating allergic diseases. Tofacitinib also inhibits, to a lesser extent, JAK1 (IC50 ¼ 100 nM) and JAK2 (IC50 ¼ 20 nM), which inhibits IFN-c and IL-6 signaling pathway and, thus, T helper type 1 (Th1) cell differentiation . Tofacitinib inhibits JAK1 and JAK3 more than JAK2; it also works by inhibiting Tyk2 to a lesser extent .
In vitro studies have shown that tofacitinib inhibits interleukin (IL)-4 dependent T helper type 2 (Th2) cell differentiation in human and mouse T cells and blocks T helper type 17 (Th17) cell differentiation. Also, the activation of STAT1 and the production of Th1 cells are inhibited. Recently Ghoreschi et al. reported that tofacitinib had been found to block more than one cytokine pathway, especially the gamma-c-cytokine signal pathway.
Tofacitinib has been used to treat T-cell-mediated autoimmune diseases such as rheumatoid arthritis, ulcerative colitis, Crohn’s disease, and psoriasis. In clinical studies, it was observed that there was a mild to moderate decrease in total lymphocyte and T cell numbers and an increase in B cell counts versus a dose-related reduction in NK cells . In heterotopic heart and kidney transplant studies, tofacitinib has shown long-term graft survival in studies on animals. Studies in heart and lung transplantation have also supported preclinical findings. A randomized, multicenter clinical renal transplant phase IIb study showed a similar proportion of acute rejection in the tofacitinib group compared to the cyclosporine-A group tofacitinib group showed better kidney function. Clinical developments in kidney transplantation have recently ceased due to worrying outcomes in the studies on the safety profile and clinical applications of the drug .
It should not be used in patients with active infection in patients with lymphocyte count less than 500/mm3, absolute neutrophil count 1000/mm3, and hemoglobin level below 9 g/dl. It should be borne in mind that the pregnancy category is C. The use of tofacitinib increases the risk of developing opportunistic infections (especially herpes zoster reactivation, pneumonia, cellulite, etc.). It also increases perforation, anemia, tuberculosis, lymphoma, non-melanoma skin cancers, and other malignancies in the diverticulitis/ gastrointestinal tract. It has been observed that the risk of EBV-associated post-transplant lymphoproliferative disease is increased when used with other immunosuppressants in patients with renal transplants.
Before starting tofacitinib treatment, it is appropriate to screen patients for latent tuberculosis, hepatitis B and C, and monitor them throughout the treatment. Also, hypercholesterolemia, anemia, and leukopenia can be seen in patients undergoing tofacitinib treatment. It should not be administered with live vaccines. In the presence of active infection, the drug should be discontinued.
In a study conducted in 2006, the use of tofacitinib has been shown to reduce intimal hyperplasia in rats undergoing aortic transplantation and prevent allograft vasculopathy . In a prospective, randomized study comparing tofacitinib and tacrolimus, there was no significant difference between the tacrolimus and tofacitinib group in terms of acute rejection rates. However, in this study, it was observed that the group receiving tofacitinib had more of the BK virus and CMV disease . In the phase IIb study of 331 renal transplant patients with low to medium risk, it was shown that there was no significant difference between tofacitinib’s high-dose (patients received tofacitinib 15 mg twice daily in 1–6 months, then 10 mg twice daily in 7–12 months) and low-dose (patients received 15 mg twice daily in 1–3 months, then 10 mg twice daily in 4–12 months) groups and Cyclosporin-A Antineoplastic and Immunosuppressive Antibiotics inhibitor structure in terms of prevention of acute biopsy. Also, patient and graft survival was similar in all groups.
Ruxolitinib
Ruxolitinib is a JAK inhibitor selective for JAK1 and JAK2 subtypes . Ruxolitinib inhibits the JAK signal associated with myelofibrosis. Its main effect is to prevent JAK from phosphorylating STAT, preventing cell division and induction of apoptosis. Studies conducted with the Oral JAK Inhibitor-I (COMFORT-I) and Oral JAK Inhibitor-II (COMFORT-II) trials have been shown to reduce spleen size and alleviate symptoms . Ruxolitinib was approved to treat medium or high-risk myelofibrosis due to positive results from COMFORT-I and COMFORT-II studies in November 2011. As a tyrosine kinase inhibitor, ruxolitinib is the first agent to be used in mycosis fungoides treatment.
According to the RESPONSE study conducted in 2014, polycythemia vera was approved when there is insufficient response or intolerance to hydroxyurea. Ruxolitinib-associated side effects include thrombocytopenia, anemia, neutropenia, increased risk of infection, and non-melanoma skin cancer. It may cause weight gain as a metabolic side effect. The drug can also cause abnormalities in alanine transaminase and aspartate transaminase levels and cause slightly elevated cholesterol levels.
Eculizumab
Eculizumab is a humanized monoclonal IgG2/4 antibody. It binds to complement C5 protein and prevents membrane attack complex formation by preventing its destruction to C5a and C5b with C5 convertase. Thus, it prevents complement-mediated cell damage . It is used to treat acute antibody-mediated rejection and thrombotic microangiopathy in kidney transplantation . It is necessary to differentiate the effects in acute and chronic antibody-mediated rejection, probably useful in acute antibody-mediated rejection but not in chronic antibody-mediated rejection. No dose adjustment is required in kidney failure; however, there is insufficient clinical data on its use in liver failure. Usually, 900 mg of eculizumab is given weekly for 4 weeks, 1200 mg weekly, then 1200 mg once every 2 weeks. Also, dose adjustment should be made in less than 40 kg .
All patients should be vaccinated against Neisseria meningitides, Haemophilus influenza, and pneumococcal pneumonia at least 14 days before application. As a result of the studies, hypertension, gastrointestinal disorders, and leukopenia are among the most common side effects associated with eculizumab .Several case reports in the literature suggest that eculizumab may be useful in the early period after HLA and ABOincompatible kidney transplants . In a study of 267 patients, the median follow-up period of the patients was 533 days, and it was found that eculizumab alone was not effective in preventing graft loss .
Another study showed that antibody-related reactions seen in the eculizumab group were much lower (7.7%, 2/26) than in the control group (41%, 21/51). In this study, no significant difference was found in post-transplant second-year allograft survival and transplant glomerulopathy . Recently, for its effectiveness in a non-blind pilot intervention study was completed regarding chronic antibody-mediated rejection. In this study involving 15 patients with kidney dysfunction (5 controls, 10 treatments), GFR was better treated in the group treated with eculizumab.
Therefore, eculizumab therapy can stabilize its function; however, more extensive studies with longer follow-up are needed to determine whether the graft function is better protected . Studies are examining the effectiveness of eculizumab in de novo in sensitized patients undergoing kidney and cardiac transplantation . According to the study’s results, as mentioned above, eculizumab can be used for antibody-mediated rejection, atypical hemolytic uremic syndrome, and thrombotic microangiopathy in patients with kidney transplantation. However, eculizumab does not affect levels of complement activating antibodies.
Lulizumab
Lulizumab pegol, an anti-CD28 monoclonal antibody, is an antagonist of T cell proliferation . Studies show that selective blocking of CD28 signals in the presence of the protected inhibitory CTLA-4 signal may be useful and fine-tuning of antigen-specific CD8b T cell responses . Accordingly, lulizumab reduces the undesirable side effects seen after transplantation, including immune reactions . Two monovalent antibodies with an antagonistic effect against CD28 as lulizumab and lulizumab-pegol are being developed . Preclinical studies of FR104, a pegylated humanized Fab fragment,and lulizumab, a pegylated anti-CD28 monoclonal antibody, show favorable results.
In an animal study, the renal allograft study demonstrated that FR104 caused enhanced immunosuppression without steroids in both low-dose calcineurin inhibitors. Selective CD28 antagonists may be an alternative calcineurin inhibitors-free strategy to B7 antagonists for kidney transplant recipients . In non-human primates transplanted with kidney allograft, low-dose tacrolimus combined with rapamycin increased long-term allograft survival . Lulizumab-pegol has been tested in non-human primates and has been shown to inhibit T cell-dependent antibody responses and cytokine production . In humans, both drugs have been evaluated in phase I clinical trials and are declared safe and well-tolerated . Therefore, a prospective multi-center study has been launched to investigate the efficacy of lulizumab in combination with glucocorticoids, belatacept, tocilizumab, and everolimus in kidney transplant recipients.
Siplizumab
Siplizumab is a humanized monoclonal antibody that acts against transmembrane CD2. CD2 antigen plays an essential role in T cell adhesion to the APC. It may also play a role in signal transduction in T cell activation . CD2 promotes adhesion of T cells to APCs through its interaction with the LFA-3 (CD58) ligand . Binding of CD2 to LFA-3 and the intracellular signals required for T cell activation, as a costimulator for this molecule . CD2 blockade has also been shown to promote islet allograft tolerance induction in a murine model . Siplizumab has demonstrated beneficial effects in preventing acute renal allograft rejection and treating acute graft-host-host-disease in a randomized study. It has been determined that siplizumab exerts potential benefits as an induction agent in renal transplanted patients, and it has an acceptable safety profile .
The anti-CD40
As known, CD40 is a cell surface glycoprotein in the TNF receptor family. CD40 is structurally expressed, and upon activation, its expression is further increased on APCs, including B cells, dendritic cells, and macrophages; CD40 expression can also be induced on parenchymal cells, such as endothelial cells and fibroblasts after inflammatory stimulation . CD40/CD40L interaction is necessary for the development of thymus-dependent humoral immune responses . Deficiency in CD40 or CD40L causes a decrease in serum IgG, IgA level, and normal/high-level IgM. This disease is also called hyper IgM syndrome, and the risk of opportunistic infections is increasing.
It is known that CD40 and CD154 are the primary targets for antigen-presenting activated T cells and play an essential role in the development of antibodies. In addition to activating the CD28:B7 stimulating pathway for modulating T-cell function, TNF-related T-cell activity also plays a role in acute and chronic allograft rejection . CD40:CD40L signalization has been evaluated in preclinical studies . The results of phase II studies in renal transplantation have been reported recently and have been shown to have higher rejection rates in the non-calcineurin group of the study. A fully humanized IgG4 antibody produced against CD40 is well tolerated at different doses ranging from 50 to 500 mg in patients undergoing kidney transplantation. Maintenance monotherapy of anti-CD40 antibody ASKP1240 for 6 months effectively suppressed both cellular and humoral alloimmune responses and prevented rejection of the hepatic allograft. For this reason, the CD40 blockade by ASKP1240 is thought to be the desired immunosuppressant for clinical liver transplantation.
CFZ533
CFZ533 is a fully human, aglycosylated immunoglobulin G1, inexhaustible anti-CD40 antibody that blocks CD154 binding to CD40 and downstream pathway activation. Recently, kidney transplantation of an anti-CD40 monoclonal antibody called CFZ533 has been performed in a nonhuman. It has been shown to prolong renal allograft survival and function when administered as monotherapy in the primate’s model. It has also been shown to be an alternative to calcineurin inhibitor treatment regimens in clinical transplantation. The results revealed that the efficacy of CFZ533 in a nonhuman primate kidney allograft transplantation model was independent of B cell depletion. The antibody was well tolerated, there was no evidence of thromboembolism or other adverse effects, and kidney function was preserved up to 100 days after transplantation in four out of five animals. It has been demonstrated that CFZ533 in kidney transplantation may be possible to prolong the kidneys’ endurance and potentially improve long-term outcomes for kidney transplant patients.
Tocilizumab
Tocilizumab is a humanized monoclonal antibody that shows activity against soluble and membrane forms of IL-6R . The FDA has approved it to treat moderate to severe rheumatoid arthritis and idiopathic juvenile arthritis . As shown before, IL-6 is a critical pleiotropic cytokine involved in stimulating B cells and plasma cells, antibody production, promoting T follicular helper cell populations, and Th17 cell differentiation. Among the most common side effects of tocilizumab in clinical studies are upper respiratory infection, nasopharyngitis, headache, and hypertension. The alanine transaminase enzyme also increased in at least 5% of patients, but in most cases without symptoms .
In another study, the use of tocilizumab therapy in 36 kidney transplant patients with chronic antibody-related reactions who failed with intravenous immunoglobulin (IVIG) and rituximab with or without plasma exchange treatment was evaluated. Tocilizumab was administered at 8 mg/kg per month with a maximum dose of 800 mg for 6–25 months. Graft and patient survival rates in patients treated with tocilizumab were 80% and 91%, respectively, at 6 years after treatment. The stabilization of renal allograft function was achieved within 2 years . No significant adverse events or serious adverse events have been reported. Tocilizumab may represent a potential alternative approach to stabilize graft function. A second phase II trial is conducted to evaluate tocilizumab’s effectiveness in reducing inflammation in the kidney allograft.
Voclosporin
Voclosporin is a new oral semi-synthetic analog of cyclosporine-A with a modification in the first amino acid residue of the molecule . It is a calcineurin inhibitor designed to treat psoriasis, prevent post-transplant organ rejection, and treat autoimmune diseases (uveitis, arthritis, type I diabetes, and Crohn’s disease) . It is declared that voclosporin has a three times stronger effect than cyclosporine-A according to in vitro results . A good correlation was observed in a study of kidney transplantation between voclosporin dose, calcineurin inhibition, and drug efficacy . According to results obtained from single-dose and multi-dose phase I studies, with the dosages of 4.5 mg/kg/day, the most frequently reported side effects were found as similar to those reported with other calcineurin inhibitors such as headache, hypertension, upper respiratory infections, and diarrhea. Also, kidney function was seen as less affected . In an animal study, low-dose voclosporin has been shown to provide similar inhibition to cyclosporine-A in lymphocyte proliferation, T cell activation, and cytokine production by the T cell. However, unlike cyclosporine-A, it does not interact with MMF.
In the phase IIb PROMISE study, in a multi-center study of voclosporin against tacrolimus, three different doses of voclosporin in addition to IL-2 receptor antagonist, MMF, and steroid therapy for 334 de novo kidney transplant patients (0.4, 0.6, or 0.8 mg/kg twice a day) or standard tacrolimus (0.5 mg/kg twice a day) were investigated. No difference in the incidence of acute biopsy-proven rejection was detected between the voclosporin and the tacrolimus group. When investigated for kidney function 6 months after transplantation, GFR was lower in the high dose voclosporin group than standard tacrolimus therapy. There was no difference between the groups in terms of hypertension and another adverse event incidence; however, it was found that the low incidence of new baseline diabetes was lower in the lowdose voclosporin group. According to the 6-month study, voclosporin is as potent as tacrolimus and similar in renal allograft function and is also thought to be associated with a reduced incidence of new-onset diabetes.
Anti-CD20 monoclonal antibody
Rituximab
Rituximab is a chimeric anti-CD20 monoclonal antibody licensed for non-Hodgkin lymphoma, chronic lymphocytic leukemia, and rheumatoid arthritis . The rituximab Fab domain binds to the CD20 antigen; the Fc domain may play a role in immune effector functions. The mechanism of action for how rituximab exerts its positive effect in the clinic is not fully understood. In kidney transplantation by itself, the administration of rituximab can lead to the rapid elimination of B cells . Besides antigen presentation, B cells perform various functions such as nucleus secretion and cytokine production. The role of TNF-a and IL-10 secreted by B cell in renal allograft damage is exact . After binding to the rituximab CD20 antigen, it affects B cells in at least three ways.
1.It stimulates the complement cascade leading to complement-mediated cytotoxicity.
2.Recognizes macrophage causing phagocytosis and antibody-dependent cell-mediated cytotoxicity.
3.It interacts with the NK cell that causes antibody-dependent cell-mediated cytotoxicity.
A decrease in B cells in peripheral blood is observed in approximately 1–3 days after rituximab administration and causes B cell depletion in most patients within 1–6 weeks. However, it has no direct effect on plasma cells . Rituximab is usually administered intravenously due to low oral bioavailability. It has been found that subcutaneous administration (bioavailability ~60%) for rituximab is not lower than IV administration in terms of pharmacokinetics and safety. The distribution volume of rituximab is 9.6 l, which indicates that it is distributed into the extravascular area but does not cross the blood-brain barrier. Rituximab has a long half-life of about 3 weeks . The initial infusion rate is started at 50 mg/h but can be increased by 50 mg/h every 30 min. The maximum infusion rate is 400 mg/h .
Side effects include flu-like symptoms, severe hypotension, and bronchospasms. If the reaction is considered mild, the infusion can be left for 1 h and then restarted in half the previous rate after the symptoms have been resolved . In a retrospective study, the rate of infectious complications was found to be 48%. In contrast, the infection rate in the control group was only 11%. While the graft survival rate was 50–60% in the control group, it was approximately 90% in the rituximab group. However, most of these studies were case series or retrospective studies. Conversely, a multicenter, randomized placebo-controlled study of the addition of rituximab in addition to standard treatment with plasmapheresis + IVIG + steroids has been conducted. In this study, it has been shown that rituximab does not have an additional benefit in 1 year on graft loss, kidney function, or DSA . More recently, a clinical study has been conducted to investigate rituximab’s efficiency in chronic transplant rejection. According to the results obtained from this study, optimized immunosuppression has been observed in the patient population to promote antidonor allogenic responses associated with positive outcomes; however, rituximab did not have such an effect.
Obinutuzumab
Obinutuzumab is a humanized, type II immunoglobulin G1 anti-CD20 monoclonal antibody. Obinutuzumab exerts its effect by binding to the type II epitope of CD20 expressed in mature B and pre-B cells as well as plasmablasts . Obinutuzumab is more effective than rituximab in depleting B cells in peripheral blood and lymphoid tissue in animal models and patients with chronic lymphocytic leukemia . Specifically, obinutuzumab caused more significant bone marrow B cell depletion than rituximab in CLL patients . In the phase 1b clinical study on obinutuzumab, the drug was well-tolerated, and most of the side effects were grades 1 and 2. A total of 11 serious adverse events occurred in 36% of patients. Among these side effects, 10 were infections, and 4 occurred after kidney transplants. IVIG treatment with obinutuzumab has been shown to cause peripheral B-cell depletion and reduce B-cells in retroperitoneal lymph nodes. Briefly, obinutuzumab has been shown to have significantly better efficacy in B-cell reduction, despite having a similar side-effect profile to rituximab .
Sotrastaurin
Sotrastaurin inhibits classical and novel protein kinase C isoforms involved in activating transcription factor NF-jB, B, T cells, and modulation of receptor-mediated signals . As a result of this mechanism of action, it blocks early T cell activation and IL-2 production. Animal studies have shown that kidney allograft prolongs survival if sotrastaurin administered as a single agent or in combination with cyclosporine-A. In the phase II study, sotrastaurin (200 mg bid) showed comparable efficacy with mycophenolic acid. After 3 months, the frequency of acute rejection increased. A higher dose of sotrastaurin combined with mycophenolic acid was shown to have a higher incidence of acute rejection than tacrolimus and mycophenolic acid.
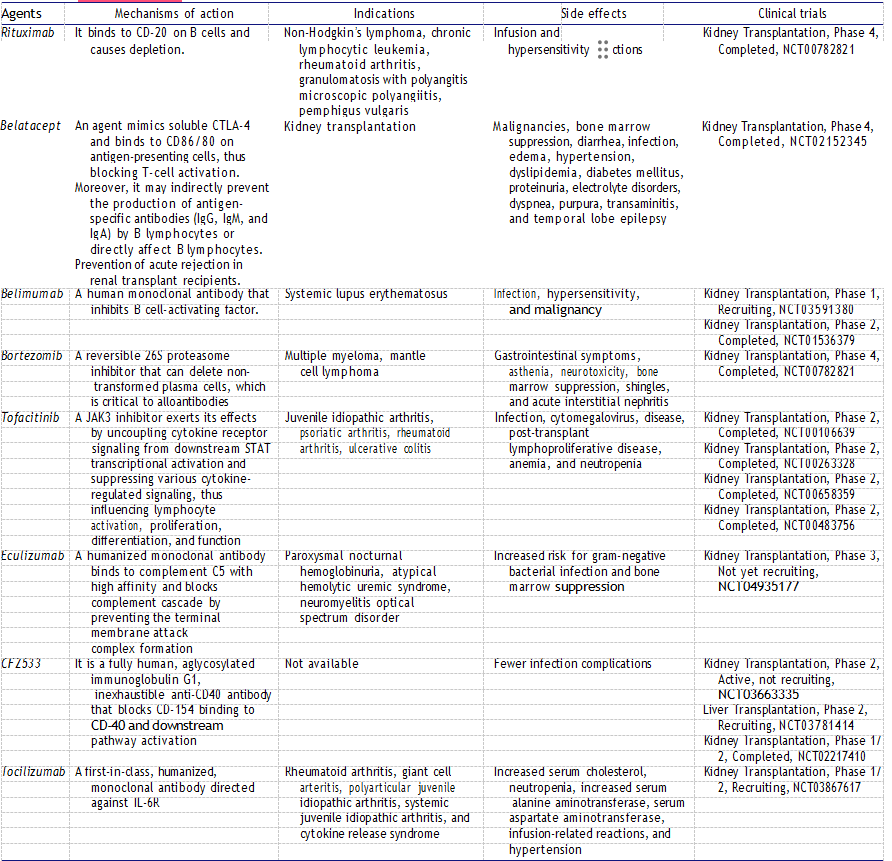
In another study, gastrointestinal and cardiac adverse events are higher in the group receiving sotrastaurin. It has also been shown that concomitant administration of sotrastaurin with everolimus causes more failure than cyclosporine-A and only improves kidney allograft function. Also, in vitro studies have shown that it can cause QT prolongation, and, therefore, another study has been conducted on healthy volunteers. According to the study, therapeutic doses have not been confirmed to cause QT prolongation. A reversible increase in average ventricular heart rate was observed in a single dose of 500 mg, and average heart rates remained within the normal range. New immunosuppressive agents, mechanism of action, indications, side effects, and clinical trials are summarized in Table 1.
Table 1. New immunosuppressive agents, mechanism of action, indications, side effects, and clinical trials.


Conclusion
Organ transplant surgeries are the curative treatment of many diseases with end-stage solid organ failure. The addition of developments in surgery in the last half of the twentieth century, especially in the field of the immune system in the 1960s, has been the basis for the extension of organ transplant surgeries to the present day. Every year, at least 100,000 solid organ transplants are carried out worldwide. Advances in the immune system have led to the broader use of immunosuppressant drugs in widespread clinical practice.
The developments in immunosuppressive drugs for the last half-century have led to the discovery of several drugs with different mechanisms of action. These developments have led to decreased tissue rejection, graft survival, and increased lifespan. Despite all these positive developments, it could not prevent critical clinical problems such as chronic allograft rejection. With careful monitoring, low doses of immunosuppression are generally well-tolerated and safe.
Therefore, immunosuppressant drugs play an essential role in maintaining the balance between tissue rejection and toxic effects. Due to the complexity, cost, and difficulty of drug development and clinical studies, clinical indicators of developments slowly develop.Despite all these difficulties, the discovery and clinical studies of many new agents continue. Preventing or limiting the adverse effects of new drugs discovered, maximizing the impact of drug combinations, and patient-specific use are among the main objectives of this field.
Currently, the most preferred immunosuppressant drugs in solid organ trans plantation and clinically significant issues, such as the mechanism of use, side effects, and new agents of new agents, have been discussed. Individualized immunosuppression therapy and the discovery of the ideal immunosuppressant drug will continue to be an essential issue for many years to come. Further experimental and clinical studies are needed to discover new agents, individualized use of immunosuppressant drugs in solid organ transplantation, increase the therapeutic efficacy, and reduce the side effects and toxicity of drugs.
