The Chemistry of 2′,3′-cGAMP: Structural Insights and the Design of Potent Analogs
Keywords: 2’3’-cGAMP, c-diGMP, CDN, cGAMP, cGAS, Chemical cGAMP analogs, Rp,Rp-2 prime 3 prime c-diAMPSS, STING
Abstract
2’3’-cGAMP formation is biocatalyzed by cGAMP synthase (cGAS) upon sensing of cytosolic double-stranded DNA (dsDNA) and functions as an endogenous inducer of innate immunity by directly binding to and activating the adaptor protein stimulator of interferon genes (STING). The cyclic dinucleotides (CDNs) cyclic diguanosine monophosphate (c-diGMP) and cyclic diadenosine monophosphate (c-diAMP) with two canonical 3 prime 5 prime internucleotide linkages are ubiquitous second messenger molecules in bacteria, regulating a multitude of physiological processes. Recently the noncanonical CDN cyclic guanosine monophosphate-adenosine monophosphate 2’3’-cGAMP featuring a mixed linkage, which consists of a 2 prime to 5 prime and a 3 prime 5 prime internucleotide bond, has been identified as a signaling molecule in metazoan species in late 2012. Thereby 2’3’-cGAMP can stimulate interferon beta (INF beta) secretion, a major signaling pathway of host defense, which is independent of toll-like receptor (TLR) activation. Medicinal chemistry of 2’3’-cGAMP and development of corresponding analogs are still in their infancy, and only a handful of structurally related compounds are available to the scientific community. The aim of this chapter is to summarize synthetic approaches to prepare canonical and noncanonical endogenous CDNs including 2’3’-cGAMP. Furthermore, we will describe syntheses of 2’3’-cGAMP analogs bearing modifications, which will facilitate further studies of the emerging biological functions of 2’3’-cGAMP and to identify additional receptor proteins. Finally, we will review latest developments concerning 2’3’-cGAMP analogs with improved hydrolytic stability in cell cultures and in tissues, putatively qualifying for new therapeutic options on the basis of 2’3’-cGAMP signaling.
Introduction
Cyclic diguanosine monophosphate, also known as cyclic bis(3 prime 5 prime) diguanylic acid (c-diGMP), was described in 1987 as an unusual cyclic dinucleotide and allosteric modulator of bacterial cellulose synthase in Acetobacter xylinum. c-diGMP consists of two guanosine-5 prime-monophosphates (5 prime-GMP) forming a 12-membered ring system that contains two phosphate diester units with canonical 3 prime 5 prime bonding. Meanwhile, c-diGMP is a well-established universal second messenger in the bacterial kingdom with multiple functions, including biofilm formation, motility, and virulence.
Cyclic diadenosine monophosphate (c-diAMP) was identified in 2008 as a signaling nucleotide involved in the regulation of DNA integrity in Bacillus subtilis during sporulation. In the following years, c-diAMP was found in multiple other bacteria and is now considered as an emerging bacterial second messenger with a steadily increasing number of reports unraveling new cellular functions in prokaryotes.
Cyclic guanosine monophosphate-adenosine monophosphate (cyclic GMP-AMP; cGAMP) with a combination of adenine and guanine nucleobases was published as the first endogenous CDN representing a second messenger function in metazoa only very recently in 2013. cGAMP is biosynthesized from ATP and GTP by cGAMP synthase (cGAS) upon sensing of cytosolic dsDNA, a typical danger signal in eukaryotic cells. Subsequently, cGAMP activates the adaptor protein stimulator of interferon genes (STING) to induce type 1 interferons and other cytokines as response of the innate immune system. Metazoan cGAMP was originally described as CDN with two canonical 3 prime 5 prime linkages between the 5 prime-GMP and 5 prime-AMP units, similar to the almost simultaneously identified bacterial 3 prime 3 prime cGAMP from Vibrio cholerae. Within some months, several labs investigated the mode of action of metazoan cGAS and characterized the mixed CDN formed with multiple methods, such as crystallography, enzymatic hydrolysis, and chemical synthesis of cGAMP isomers with defined chemical structures. Thereby it was confirmed that in metazoan cGAMP the internucleotide phosphate-binding motifs constituted of a quite uncommon combination of a noncanonical 2 prime 5 prime linkage together with a canonical 3 prime 5 prime linkage 2’3’-cGAMP. The phosphodiester-containing 12-membered ring system in 2’3’-cGAMP is at least theoretically rather flexible and can adopt the syn or anti conformation with respect to the two nucleobases adenine and guanine. Also the orientation of each nucleobase toward its dedicated ribose can exist in syn and anti conformation as a result of free rotation around the glycosidic bond. From all hypothetical conformers, in crystals of 2’3’-cGAMP bound to the homodimeric STING protein, the nucleobases adenine and guanine are oriented syn to each other and anti to their corresponding ribose unit.
There is dramatically increasing evidence that metazoan 2’3’-cGAMP with its immunomodulatory activity can serve as lead for a novel therapeutic paradigm as adjuvant for vaccines or to combat cancer, HIV, and other major diseases related to pathogenic viruses, bacteria, and protozoa. The scope of this chapter is to provide a short overview on synthetic routes for the design of canonical and noncanonical CDNs, like 2’3’-cGAMP and related structures. Furthermore, we will describe syntheses which introduce chemical modifications into noncanonical CDNs. The resulting analogs, featuring modulated biological activities, are versatile chemical tools to further investigate the cGAS-2’3’-cGAMP-STING pathway and beyond. Lastly, we will summarize very recent efforts of preclinical testing with 2’3’-cGAMP analogs in cancer pharmacology that may translate into early clinical development in the near future.
Syntheses of Canonical CDNs and 2’3’-cGAMP
Over the last 30 years, several syntheses of c-diGMP and c-diAMP have been published, applying phosphate, phosphonate, and phosphite chemistries in solution phase. The majority of these synthetic protocols rely on nucleoside precursors with suitable protection groups to allow for regiospecific reaction steps. Several of these protocols were also applied for the synthesis of CDNs bearing two heterogenic nucleobases, like guanine and adenine for the generation of 3 prime 3 prime cGAMP. In general, this nucleoside approach is convenient for the preparation of small collections of CDNs differing in their nucleobase composition. However, it should be emphasized that most of these protocols include more than 10 synthetic steps accompanied by several chromatographic purifications. In the final CDN the nucleoside combination is defined by the initial selection of nucleoside precursors. This implies that each synthetic process is only suitable to prepare a single cyclic dinucleotide with a unique nucleobase signature. To this end, all these synthetic procedures are not compatible with combinatorial chemistry strategies, a severe drawback limiting the access to a large number of CDN analogs. This obstacle was tackled at least in part by Giese and coworkers with the preparation of a common cyclic ribose-3 prime 5 prime-3 prime 5 prime-diphosphate building block that was fused with a nucleobase only at the end of the multistep reaction.
As it stands for now, this improved flexibility of the backbone approach was reported only for the preparation of CDNs with identical nucleobases, including c-diGMP and c-diAMP. The generation of heteromeric CDNs should be possible by employing a 1:1 mixture of two distinct nucleobases in the final ribosylation step. However, subsequent separation and purification of heteromeric CDNs and homomeric by-products by HPLC are expected to be challenging and might be prohibitive for a scale-up of this method. In the future, a further elaborated backbone approach might qualify for combinatorial strategies to fuse a large number of preactivated nucleobases with a universal cyclic backbone intermediate for the generation of CDN libraries.
While all these reactions are based on solution chemistry, Sintim and coworkers reported a solid-phase approach for the preparation of c-diGMP using a DNA synthesizer and two different guanosine phosphoramidites, including 5 prime DMTr-2 prime O-TBDMS-guanosine phosphoramidite, carrying a methyl phosphate protection group. In this protocol, the cyclization step with 1-(mesitylene-2-sulfonyl)-3-nitro-1,2,4-triazole (MSNT) in pyridine to prepare the protected CDN was critical with only moderate yields below 50%. Subsequently, in a second-generation approach, the authors cleaved off the protected linear dimer from the solid support prior to quantitative cyclization with MSNT in solution.
Strobel and coworkers further developed syntheses in heterogenic phase making use of the most common and commercially available nucleoside phosphoramidites with 5 prime DMTr, 2 prime O-TBDMS protecting groups, as well as 3 prime O-[(2-cyanoethyl)-N,N-diisopropylaminophosphinyl] functionalization. Quantitative cyclization to generate the protected CDN was accomplished on bead by repeated prolonged treatment with MSNT in pyridine combined with intermittent pyridine washing steps. As mostly automated syntheses, these solid support methods could be advantageous to solution chemistry to prepare multiple CDNs in a shorter time frame. They could therefore represent a good starting point for effective scale-up and process development of CDN synthesis in the future.
Large-scale and cost-saving syntheses of CDNs are eagerly awaited by the scientific community to have at command sufficient quantities of these second messengers for detailed biological and pharmacological testing. To meet these demands Jones and coworkers developed a solution-phase method, which gives access to c-diGMP as well as related phosphorothioate analogs in a gram scale and is currently considered as state of the art. It uses the common and commercially available precursor (N2-iBu-5 prime O-DMTr-2 prime O-TBDMS-3 prime O-CEP-guanosine) and is based on phosphoramidite phosphonate chemistry protocols established before by this laboratory. The innovation, however, lies in a carefully chosen synthetic route, applying reaction conditions that allow for the whole process to be performed in a single-flask synthesis without the need for any intermediate purification steps. This improved protocol thus demonstrates a very straightforward and scalable method. Furthermore, after the final deprotection, c-diGMP is simply crystallized from the reaction mixture, avoiding cost-intensive chromatographic purifications throughout the whole reaction sequence. Also the cyclic diguanosine monophosphorodithioate isomers Rp,Rp-c-diGMPSS and Rp,Sp-c-diGMPSS were prepared with this improved method employing sulfurization instead of oxidation reagents.
Here, only a single purification step for the separation of Rp,Rp and Rp,Sp isomers is necessary. In these dithiophosphates of c-diGMP, the terms Rp and Sp refer to the different isomers of the sulfur-modified and thus chiral phosphorus atoms of the 12-membered ring system of cyclic dinucleotides according to the Cahn-Ingold-Prelog priority rules (CIP). The isomer Sp,Sp-c-diGMPSS is chemically not accessible by this synthetic protocol as a result of the stereoselective introduction of sulfur into the phosphonate intermediate with 3-H-1,2-benzodithiol-3-one.
Shortly after the first reports of metazoan cGAMP, Jones and coworkers adapted their single-flask c-diGMP protocol to be applicable for the synthesis of mixed cyclic dinucleotides containing adenine and guanine. With the modified procedure in hands, they succeeded in preparing several cGAMP isomers with canonical and noncanonical linkages. In particular, to facilitate the 2 prime 5 prime linkages, they interchanged the 2 prime and 3 prime functionalities of the second precursor. The synthetically prepared and structurally defined 2’3’-cGAMP, applied as HPLC reference, helped to confirm the chemical identity of native metazoan cGAMP. Meanwhile, this single-flask protocol was successfully established in other laboratories, partially modified and used to prepare 2’3’-cGAMP and analogs in larger scale.
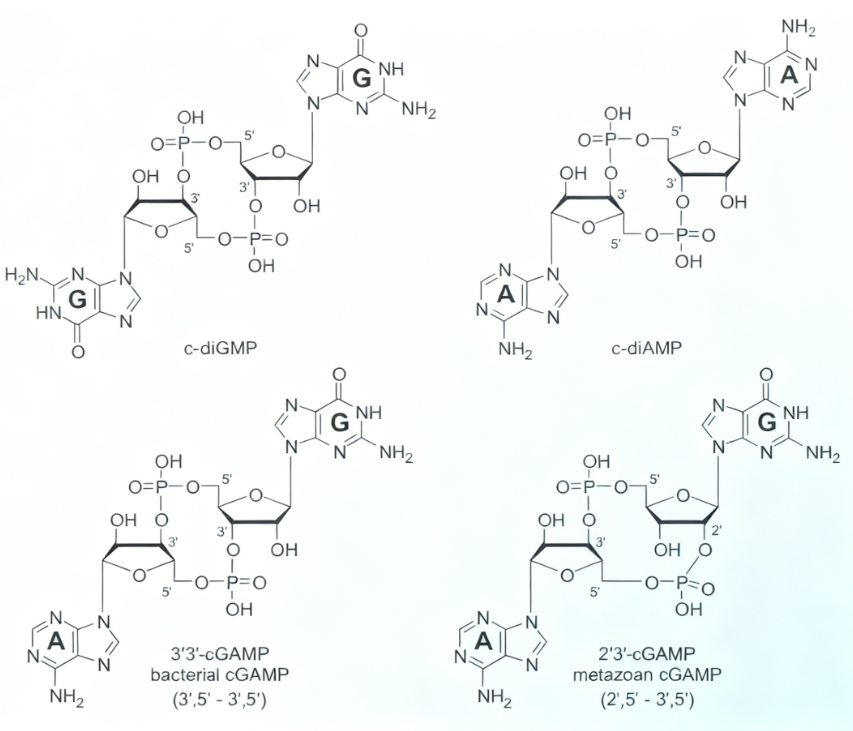
Figure 1 Canonical and noncanonical natural cyclic dinucleotides (CDNs). All structural drawings in this chapter depict the free acid form of the phosphate moieties in the 12-membered ring system. At physiological pH each of the phosphate diesters is negatively charged. The figure shows chemical structures of c-diGMP, c-diAMP, 3 prime 3 prime cGAMP (bacterial cGAMP with 3 prime 5 prime to 3 prime 5 prime linkage), and 2’3’-cGAMP (metazoan cGAMP with 2 prime 5 prime to 3 prime 5 prime linkage).
Syntheses of 2’3’-cGAMP Analogs
Analogs with Modifications in the Nucleobase and Ribose Moieties
2’3’-cGAMP analogs with meaningful chemical modifications are thought to be useful tools for a more detailed insight into the recently unraveled cGAS-2’3’-cGAMP-STING pathway. They can also support the identification of additional receptor proteins in this emerging scientific field of noncanonical cyclic dinucleotides in eukaryotes. We set out to generate 2’3’-cGAMP analogs with functional spacers in different positions of the molecule to be employed for interaction studies with STING and for a chemical proteomics approach to search for additional binding proteins. For this task, we selected position 8 of the guanine moiety and the 2 prime OH and 3 prime OH groups of the ribose parts of 2’3’-cGAMP for a first generation of chemical interventions. Regioselective introduction of bromine into position 8 of guanine was accomplished by reacting 2’3’-cGAMP with bromine in aqueous solution at ambient temperature. Under these conditions only guanine, but not the equivalent position in adenine, is susceptible to bromination, leading to 8-Br-2’3’-cGAMP, the first 2’3’-cGAMP analog with a single modification in the nucleobase.
Nucleophilic substitutions of 8-bromoguanine-containing nucleosides and nucleotides with standard aminoalkylamino reagents are only possible in sealed containers under harsh conditions at temperatures of up to 160 degrees Celsius. The alternative thiol groups in omega-aminoalkylthiol reagents are better nucleophiles and commonly used to connect terminal aminoalkyl spacers with thioether linkage to position 8 of guanine. Therefore, 8-Br-2’3’-cGAMP was further reacted with aminoethylthiol (AET) under alkaline conditions at moderate temperatures (50 to 70 degrees Celsius) to yield cyclic 8-AET-2’3’-cGAMP. Introduction of hexyl spacers with a terminal amino group into 2 prime and 3 prime positions of 2’3’-cGAMP was accomplished according to Hammerschmidt et al. by initial activation of the lipophilic bis-diisopropylethylammonium salt of 2’3’-cGAMP with 1,1 prime carbonyldiimidazole under in anhydrous DMF, followed by addition of excess 1,6-diaminohexane. The raw mixture of 2 prime AHC-2’3’-cGAMP and 3 prime AHC-2’3’-cGAMP was purified by HPLC to isolate the spacer-modified 2 prime and 3 prime isomers of 2’3’-cGAMP. All analogs with functionalized spacers can serve as precursors for further chemical modifications, including immobilization on agarose beads, coupling to chips for surface plasmon resonance experiments, and labeling with reporter groups like biotin or fluorescent dyes.
Furthermore, after coupling to haptens these functionalized 2’3’-cGAMP analogs can be suitable tools for antibody generation and subsequent development of immunoassays for 2’3’-cGAMP. Such immunoassays could be employed to simplify the detection and quantification of 2’3’-cGAMP in biological matrices. Currently, the state of the art is still highly sophisticated HPLC-MS/MS analytics, which has sufficient detection sensitivities, but depends on high-end HPLC-MS hardware, precluding its broad application as routine method.
Immobilization on agarose beads was initiated by reacting the spacer-containing 2’3’-cGAMP analogs with N-hydroxysuccinimide ester (NHS)-activated agarose in anhydrous DMSO in the presence of a non-nucleophilic base like diisopropylethylamine. Potential applications of the resulting tools 8-AET-2’3’-cGAMP-agarose, 2 prime AHC-2’3’-cGAMP-agarose, and 3 prime AHC-2’3’-cGAMP-agarose are not only affinity purifications of STING, but also the screening for additional interaction partners of STING, which should co-elute during chromatography or co-precipitate in pulldown experiments. Furthermore, these agaroses could be employed in the quest for new binding proteins of 2’3’-cGAMP in metazoa.
In this regard, it is of interest that CDNs, including 2’3’-cGAMP, have been reported to bind to a newly identified C-linker binding pocket (CLP) in the cytosolic C-linker region of the hyperpolarization-activated cyclic nucleotide-gated channel 4 (HCN4), the main isoform of the cardiac pacemaker. HCN4 is activated by hyperpolarization of membrane voltage in myocytes, and its open probability is controlled and prolonged by cAMP binding to the cyclic nucleotide-binding domain, which is separate from the CLP site. Upon binding to CLP, 2’3’-cGAMP and bacterial CDNs were shown to antagonize cAMP regulation of the channel leading to an efficient reduction of heart rate by 30%. This first report about a putative signaling role of 2’3’-cGAMP apart from the innate immunity system highlights that physiological roles of 2’3’-cGAMP in mammals are far from being fully evaluated, including the existence of additional protein-binding partners that may be identified by the chemical proteomics approach with 2’3’-cGAMP-agaroses as affinity baits.
Biotinylated analogs of the canonical bacterial second messengers c-diGMP and c-diAMP were synthesized by Beaucage and coworkers and in our laboratory. Biotinylated c-diGMP analogs were shown to interact with certain previously established c-diGMP binders and, after coupling to streptavidin beads, were suitable to isolate putative c-diGMP interacting proteins from murine macrophages after challenge with Legionella pneumophila. 2 prime [Biotin]-AHC-c-diAMP was used to develop a c-diAMP ELISA and after immobilization on streptavidin-coated magnetic beads, employed for the identification of new receptor proteins from Staphylococcus aureus and Bacillus subtilis in pulldown assays. To enable comparable studies in the field of metazoan 2’3’-cGAMP, the omega-aminospacer-containing analogs 8-AET-2’3’-cGAMP, 2 prime AHC-2’3’-cGAMP and 3 prime AHC-2’3’-cGAMP were reacted with biotinamidocaproate-NHS ester ([Biotin]-NHS) in borate buffer pH 9 and 2-propanol. Raw mixtures were subsequently purified with ion exchange and reversed phase chromatography to remove unreacted starting material and hydrolyzed biotinamidocaproic acid contaminations, leading to 8-[Biotin]-AET-2’3’-cGAMP, 2 prime [Biotin]-AHC-2’3’-cGAMP, and 3 prime [Biotin]-AHC-2’3’-cGAMP, respectively.
As was shown for the biotinylated derivatives of bacterial second messengers in recent years, these new biotinylated noncanonical congeners are expected to serve as valuable tools for further characterization of 2’3’-cGAMP signaling in eukaryotes.
The fluorescein-labeled analog 2 prime Fluo-AHC-c-diGMP is commercially available and was employed to detect c-diGMP-binding proteins in c-diGMP-agarose eluates, thereby avoiding radioactive assays. This compound was also used to develop a fluorescence polarization-based assay to measure binding affinities of flavonoid-based agonists of mouse STING that compete for the CDN binding site. Very recently, 2 prime Fluo-AHC-c-diGMP was applied to determine binding affinities of c-diGMP to receptor proteins with microscale thermophoresis coupled to fluorescence detection. 2 prime MANT-c-diGMP and 2 prime 2 double prime Di-MANT-c-diGMP containing the fluorescent and environmentally sensitive N-methylanthranilic (MANT) dye were used for the characterization of c-diGMP-binding proteins and for staining of putative c-diGMP-binding partners in cytosolic vesicles of macrophages after Legionella pneumophila challenge. Given the variety of potential applications for fluorescently labeled 2’3’-cGAMP, we prepared a first series of fluorescein-containing analogs. For this purpose, we reacted 8-AET-2’3’-cGAMP, 2 prime AHC-2’3’-cGAMP, and 3 prime AHC-2’3’-cGAMP with 5-carboxyfluorescein-NHS ester in borate buffer pH 9 and DMSO. The reaction products 8-Fluo-AET-2’3’-cGAMP, 2 prime Fluo-AHC-2’3’-cGAMP, and 3 prime Fluo-AHC-2’3’-cGAMP were purified by repeated reversed phase chromatography to remove fluorescent impurities. The synthesis of MANT and di-MANT-substituted 2’3’-cGAMP and other related fluorescent analogs are ongoing projects in our laboratory.
Syntheses of 2’3’-cGAMP Analogs with Modifications in the Phosphate and Ribose Moieties
In 2014, Mitchison and coworkers identified ectonucleotide pyrophosphatase phosphodiesterase 1 (ENPP1) as dominant endogenous hydrolase of 2’3’-cGAMP in an activity-guided screening and protein purification sequence from calf liver. ENPP1 belongs to the ENPP family consisting of three members (ENPP1 to 3) and is described as a membrane-associated glycoprotein with an extracellular active site of high ATPase activity. Furthermore, ENPP1 can be released into serum and exists as a functional soluble enzyme. The protein has only weak substrate specificity and cleaves a variety of pyrophosphate bonds in nucleotides, including ATP, ADP, NAD+, ADP-ribose, FAD, CoA, and diadenosine polyphosphates, leading to 5 prime AMP. Also UDP-glucose and other pyrimidine nucleotides may serve as substrates. In incubations with recombinant ENPP1, 2’3’-cGAMP is hydrolyzed efficiently with similar kinetics as the natural substrate ATP. Given this apparent instability of the natural metazoan second messenger, the bacterial 3 prime 3 prime cGAMP and a set of 2’3’-cGAMP analogs with modifications in the ribose or the phosphate moiety were synthesized and tested for improved hydrolytic resistance against ENPP1. 3 prime 3 prime cGAMP was prepared chemoenzymatically from ATP and GTP by dinucleotide cyclase (DncV), an endogenous cyclase from Vibrio cholerae. All noncanonical CDNs were enzymatically synthesized with cGAS, which was activated by DNA from herring testes (HT DNA), and appropriately modified ATP and GTP analogs. Incubation of 3 prime deoxy-GTP with ATP resulted in 3 prime deoxy-2’3’-cGAMP, which showed a half-life of approximately 1 hour in ENPP1 hydrolase assays, comparable to 2’3’-cGAMP itself. By contrast, the mono and dithiophosphate analogs 2 prime 3 prime cGSAMP, 2 prime 3 prime cGASMP with sulfur either in the 2 prime 5 prime or 3 prime 5 prime linkage and 2’3’-cGAMPSS (2 prime 3 prime cGSASMP) demonstrated significantly improved hydrolytic stability against ENPP1. The half-life of all thiophosphate-containing CDNs was in the range of 24 hours or even longer. This is in accordance with former reports about increased biological stabilities of sulfur-containing nucleoside phosphorothioates compared to unmodified phosphodiesters in cyclic nucleotides, triphosphates, or oligonucleotides. 2 prime 3 prime cGSAMP, 2 prime 3 prime cGASMP, and 2’3’-cGAMPSS were prepared by incubation of cGAS with ATP, GTP, ATP-alpha-S, and GTP-alpha-S in different combinations. In this particular chemoenzymatic protocol, diastereomeric mixtures of Rp and Sp-ATP-alpha-S and GTP-alpha-S were used for the biocatalyzed syntheses, and the reaction products were not fully characterized with respect to their diastereomeric composition of distinct isomers. A careful evaluation of both potentially stereoselective events, the enzymatic substrate recognition and the cyclization step by cGAS, is thought to be possible with commercially available pure Rp and Sp isomers of ATP-alpha-S and GTP-alpha-S. 3 prime deoxy-2’3’-cGAMP and 2’3’-cGAMPSS were shown to bind to human STING with high affinities in the low nanomolar concentration range, comparable to endogenous 2’3’-cGAMP. Subsequently, these analogs were further tested against 2’3’-cGAMP for their ability to activate human STING in cell culture in long-term incubations of 24 hours. Of all analogs tested, the nonhydrolyzable 2’3’-cGAMPSS exhibited highest levels of INF beta induction in human THP-1 cells expressing STING. The effective concentration (EC50) was approximately 5 micromolar for 2’3’-cGAMPSS, approximately tenfold lower compared to 2’3’-cGAMP. From additional experiments with lung fibroblast cells from Enpp1 mice with similar EC50 values for both CDNs, the authors concluded that the improved EC50 value of 2’3’-cGAMPSS was due to increased biostability compared to 2’3’-cGAMP. In summary, 2’3’-cGAMPSS was recognized as superior lead for further therapeutic development. Interestingly, also the bacterial 3 prime 3 prime cGAMP was found to be stable against hydrolysis by ENPP1. Albeit 3 prime 3 prime cGAMP had a 50 to 100-fold reduced binding affinity toward human STING in vitro, its improved hydrolytic stability resulted in similar activatory potentials of 3 prime 3 prime cGAMP and 2’3’-cGAMP, with EC50 values of approximately 50 micromolar for INF beta induction in human THP-1 cells. These initial results opened up another track for future medicinal chemistry interventions to develop CDN-based STING activators with prolonged stability in biological systems.
Applying the general chemical sequence reported by Gaffney et al., a series of phosphate-based and phosphorothioate-modified CDNs with canonical and noncanonical linkages have been prepared and tested for their biological activity. Besides phosphate-containing CDNs, also the pure isomers Rp,Rp-2’3’-cGAMPSS, Rp,Sp-2’3’-cGAMPSS, Rp,Rp-2 prime 3 prime c-diAMPSS, Rp,Sp-2 prime 3 prime c-diAMPSS, Rp,Rp-2 prime 3 prime c-diGMPSS, and Rp,Sp-2 prime 3 prime c-diGMPSS were synthesized from commercially available nucleoside phosphoramidites and purified by HPLC.
The resulting analogs were tested for binding to and activation of STING wild type (WT) and four different haplotypes of human STING protein genotypes (REF, HAQ, AQ, and Q alleles), varying in amino acid positions 71, 230, 232, and 293. These single nucleotide polymorphisms were reported to affect innate immune signaling. As reported earlier, highest binding affinity for wild type human STING was achieved by analogs with noncanonical linkage, namely 2’3’-cGAMP, Rp,Rp-2’3’-cGAMPSS, and Rp,Rp-2 prime 3 prime c-diAMPSS. For activity screening each of the five STING variants was stably expressed in HEK293T cells, which are negative in endogenous STING. Induction of INF beta was monitored by co-transfection of a IFN beta-luciferase construct. Bacterial CDNs c-diGMP, c-diAMP, and 3 prime 3 prime cGAMP were able to induce IFN beta in genotypes hSTING WT, hSTING HAQ, and hSTING AQ, but not in hSTING REF and hSTING Q. By contrast, the noncanonical endogenous 2’3’-cGAMP, Rp,Rp-2’3’-cGAMPSS, Rp,Rp-2 prime 3 prime c-diAMPSS, and Rp,Rp-2 prime 3 prime c-diGMPSS were able to induce an IFN response in all STING variants tested. In an alternative protocol, the murine bone marrow-derived dendritic cell line DC2.4 was used for INF beta induction and to compare noncanonical 2 prime 3 prime c-diAMP with its corresponding diphosphorothioate congeners Rp,Rp-2 prime 3 prime c-diAMPSS and Rp,Sp-2 prime 3 prime c-diAMPSS. Highest levels of INF beta were detected after treatment with Rp,Rp-2 prime 3 prime c-diAMPSS. The isomeric Rp,Sp-2 prime 3 prime c-diAMPSS generated only a moderately increased INF beta level, similar to 2 prime 3 prime c-diAMP. STING signaling was further evaluated with canonical and noncanonical CDNs in peripheral blood mononuclear cells (PBMC) from human donors with different STING genotypes. In these assays the diphosphorothioated analogs Rp,Rp-2’3’-cGAMPSS, Rp,Rp-2 prime 3 prime c-diAMPSS, and Rp,Rp-2 prime 3 prime c-diGMPSS induced significantly higher levels of INF beta than canonical or noncanonical CDNs without sulfur modification. As a consequence of their superior and pan-genetic activity, Rp,Rp isomers of diphosphorothioated noncanonical CDNs were selected for further preclinical development and tested in mice bearing different solid tumor models. Upon intratumoral injection Rp,Rp-2 prime 3 prime c-diAMPSS not only significantly reduced tumor growth with a higher therapeutic index compared to Rp,Rp-2 prime 3 prime c-diGMPSS but also induced a profound systemic immune response eligible to combat metastases distant to the injection site. Importantly, the formulation of Rp,Rp-2 prime 3 prime c-diAMPSS as adjuvant together with a granulocyte-macrophage colony-stimulating factor (GM-CSF) secreting tumor cell vaccine (STINGVAX) produced a strongly enhanced antitumor response in several solid tumor models in mice. This pharmacological effect was significantly higher when compared with the vaccine alone, formulations with canonical 3 prime 3 prime c-diAMP, or formulations with other control adjuvants. In conclusion, Rp,Rp-2 prime 3 prime c-diAMPSS was selected as the most promising and first noncanonical CDN for further clinical development as potential anticancer medicine.
Conclusion and Future Perspectives
The noncanonical CDN 2’3’-cGAMP was identified as an endogenous small second messenger molecule in metazoa in late 2012. It soon became clear that 2’3’-cGAMP has a central function in innate immunity by activating STING leading to subsequent induction of IFN beta, a major pathway to combat cancer or other severe diseases caused by infections with pathogenic bacteria, protozoa, and viruses. Fortunately, a fast adaption of synthetic protocols originally established for canonical CDNs to 2’3’-cGAMP was possible with commercially available nucleoside phosphoramidite precursors. Thereby sufficient amounts of 2’3’-cGAMP were made available for intensive biological testing of the cGAS-2’3’-cGAMP-STING pathway in several laboratories.
2’3’-cGAMP was modified in different positions (C-8 of guanine, 2 prime OH and 3 prime OH of riboses) with functional spacers eligible for coupling to agarose beads or for the introduction of reporter groups, like biotin, digoxigenin, and fluorescent dyes. This new toolkit is thought to facilitate the identification of so far unknown STING or ENPP1-interacting proteins and the quest for new primary receptors of 2’3’-cGAMP signaling. New 2’3’-cGAMP analogs with reporter groups will enable an in-depth biochemical characterization of protein-CDN interactions, as was already proven successful with corresponding bacterial CDNs. 8-Fluo-AET-2’3’-cGAMP, 2 prime Fluo-AHC-2’3’-cGAMP, and 3 prime Fluo-AHC-2’3’-cGAMP are molecular probes that may be used to establish fluorescence-based binding assays for 2’3’-cGAMP and analogs at receptor proteins. Moreover, analogs like 8-AET-2’3’-cGAMP, 2 prime AHC-2’3’-cGAMP, and 3 prime AHC-2’3’-cGAMP are convenient starting points for the development of antibodies against 2’3’-cGAMP and for immunoassays to simplify the quantification of 2’3’-cGAMP in biological systems like cells and tissues.
Diphosphorothioate-modified noncanonical CDNs were shown to have significantly improved stability against hydrolysis by ENPP1 in vivo combined with preserved binding affinities and activatory potential for several genotypes of human STING protein. From this class of sulfur-containing CDNs, Rp,Rp-2 prime 3 prime c-diAMPSS exhibited pharmacological characteristics justifying further preclinical development work and putative transition into the clinical phase within only 3 years after the identification of 2’3’-cGAMP. In this context, it will be of high importance to create sophisticated analogs of Rp,Rp-2 prime 3 prime c-diAMPSS or related CDNs with modifications in multiple positions of their unique chemical architecture to fully evaluate the pharmacological potency of these new therapeutic lead structures.
Despite enormous achievements that have been accomplished thanks to intensive research during the last years, it should be emphasized that larger collections or libraries of differently modified analogs of 2’3’-cGAMP are only hardly accessible with currently established chemical methods. All nucleoside approaches require a minimum of eight chemical steps, rely on available preformed nucleoside phosphoroamidites with several protection groups, and yield only a single or two CDN analogs. It should be noted that most nucleobase modifications or ribose modifications have to be introduced on the nucleoside level often via multiple chemical steps prior to formation of the nucleoside phosphoroamidite. In addition, such modifications have to withstand and should not interfere with reaction conditions of subsequent CDN formation. All these framework conditions are bottlenecks and prohibit the synthesis of libraries of CDN analogs in the midterm. A modification of the antipodal backbone approach with alternative common cyclic ribose-2 prime 5 prime-3 prime 5 prime-diphosphate or cyclic ribose-2 prime 5 prime-3 prime 5 prime-diphosphorothioate building blocks could be a practicable starting point for the development of parallel syntheses or combinatorial chemistry procedures to generate a larger number of nucleobase-modified noncanonical CDNs. As long as this general drawback has not been overcome by innovative chemical approaches, the design and preparation of new 2’3’-cGAMP analogs will not pick up pace but will require multiple step protocols to obtain single compounds.
